Caranx Medical announces world-first successful robotic TAVI procedure
Caranx successfully carried out the procedure on a porcine model of a body in a bid to make the procedure more widespread and available.
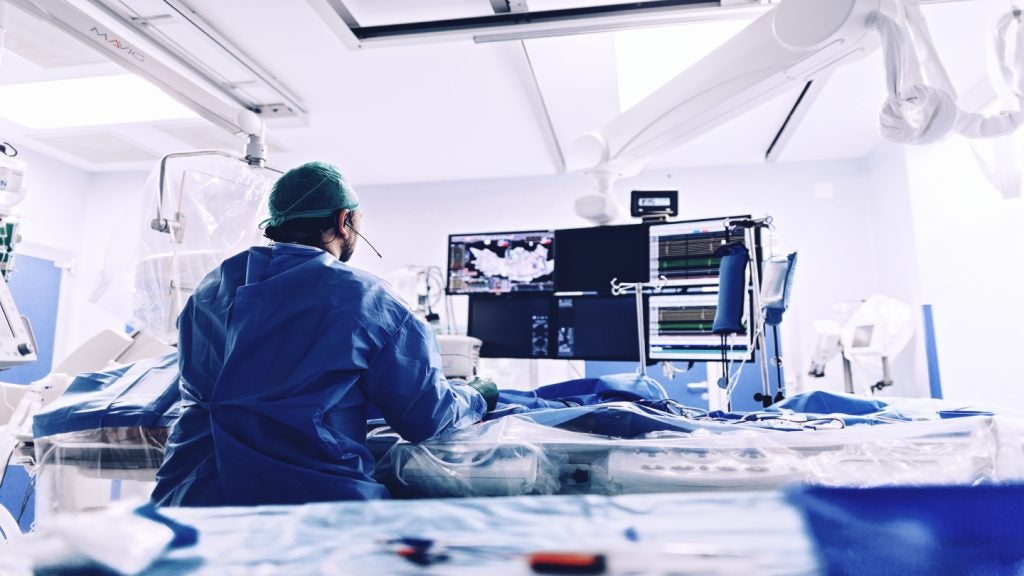
Caranx successfully carried out the procedure on a porcine model of a body in a bid to make the procedure more widespread and available.

The study will assess the economic and healthcare outcome benefits of Acarix's AI-powered CADScor System compared to traditional stress test.
The medical devices industry continues to be a hotbed of patent innovation. Activity is driven by increased need for homecare,...


Medical Technology is our digital magazine, free to read online on all devices. Click the magazine cover to read the latest issue. You can subscribe for free to have each new issue delivered to your inbox.
MMI secures FDA approval for Symani Surgical System
Cleerly touts new data for AI cardiovascular software
Avicenna.AI wins two FDA clearances for AI tools in CT scans
World Air Quality Report: what are the health impacts and where is it worst?