Cell and gene therapy company ThermoGenesis and its joint venture ImmuneCyte have launched a comprehensive suite of Covid-19 products, combining diagnostic, therapeutic and service offerings to detect, track and fight the SARS-CoV-2 virus.
The companies are developing a number of antigen testing products, including reverse transcription polymerase chain reaction (RT-PCR) tests, to complement the SARS-CoV-2 IgM/IgG Antibody Test Kit, which has been granted Emergency Use Authorization (EUA) by the US Food and Drug Administration (FDA). The joint venture also plans to offer laboratory services to all its clients, acting as a one-stop shop for all testing needs.
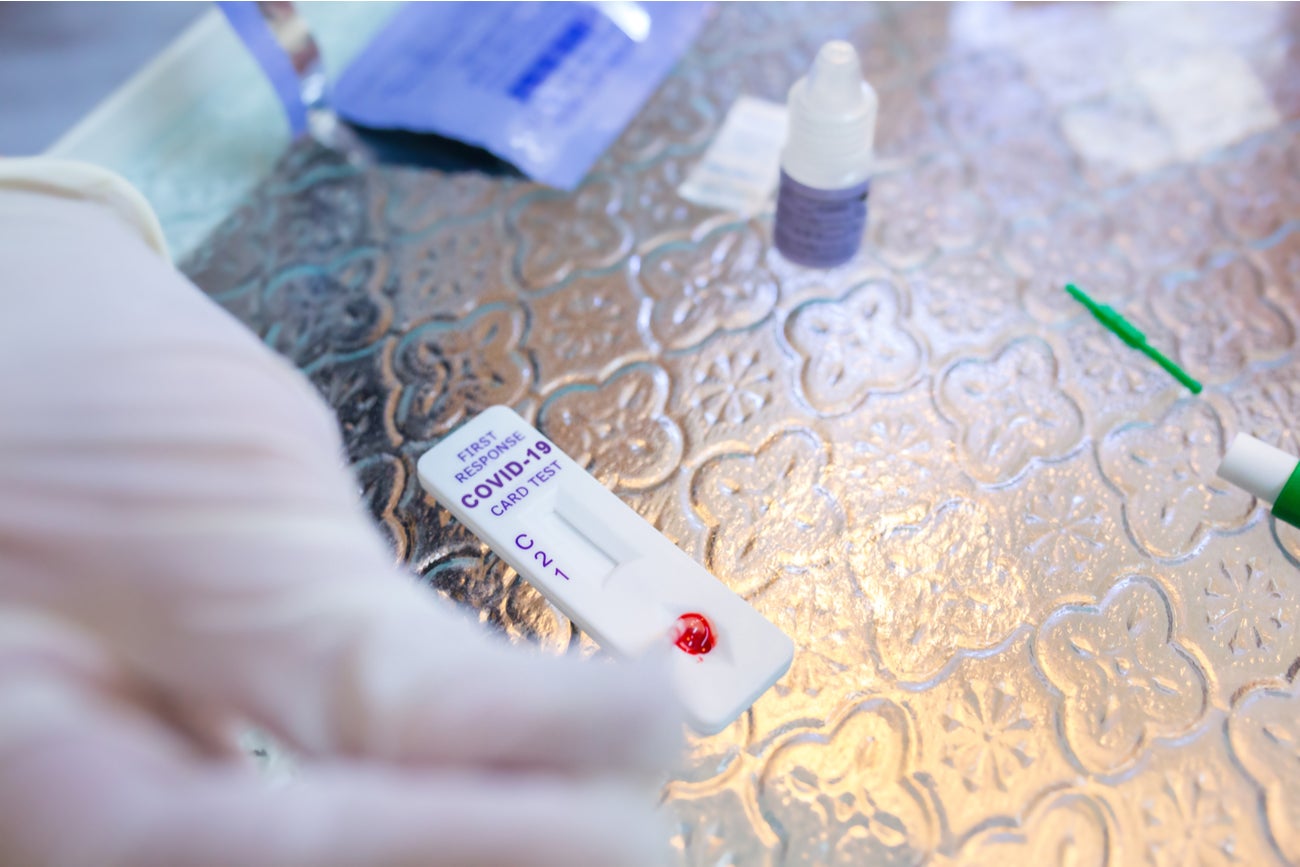
The SARS-CoV-2 IgM/IgG Antibody Test Kit is a single-use rapid immunochromatographic test for the detection of IgM and IgG antibodies in human serum, plasma and whole blood. The antibodies within the sample react with a SARS-CoV-2 recombinant N-protein antigen, which is bound to gold particles within the device. Much like a pregnancy test, these particles react with the sample and prompt the device to show two or three bars if positive, depending on the types of antibodies detected, and one if negative, showing that there are no antibodies in the sample.
Alongside its diagnostic portfolio, the organisations are working on a therapeutic pipeline for the disease. Verdict Medical Devices speaks to ThermoGenesis CEO Chris Xu about the company’s aspirations going forward.
Chloe Kent: Can you walk me through the rapid antibody test kit that ThermoGenesis has been working on?
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataChris Xu: We are taking a common platform that people are very familiar with. It’s the same platform developed for pregnancy tests and some drug abuse tests. We are using that platform to develop a rapid antibody test against Covid-19.
It takes only two or three minutes, just like a pregnancy test, and it needs only one droplet of blood. The intention of having this is so that we can do massive population screenings, to see as a society how broadly this virus has reached out and how many of us have developed an antibody response toward the virus.
CK: What is ThermoGenesis’s USP?
CX: As a medical device company we certainly see the larger operation. Epidemiologists would like to see what percentage of people test positive for antibodies in wider areas like London or New York. So we’ve also come up with a more precise portable reader for the device that can give more a digitised, quantified readout, as far as the relative amounts of antibodies in people against the virus and whether that changes over time. So we address some of the more sophisticated questions with the companion reader.
But that’s really for high-throughput labs. This test doesn’t require the reader and you can do it at home, although under FDA regulation some kind of medical intervention is required. But really, the assay is designed to be used in the long run just like a pregnancy test but for Covid-19; maybe you can get it from a local pharmacy shop and read it at home.
As a therapeutic company, we can then take this a step further. If we have people already show that they have produced antibodies, then we can generate antibody therapeutics either by asking people to donate their plasma or by banking their immune cells and storing them for the donor.
CK: As in CAR-T therapy?
CX: CAR-T therapy is a marvellous way to use somebody’s own cells to drive any cell therapy against cancer. The clinical data was just stunning and really led to a global rush for really every medical institution to get into. It think that’s going to revolutionise our future treatment against cancer. However, with a caveat – you need to harvest your own cells for future treatment of cancer.
How does that tie into Covid-19? Now we’re in the process of providing a service where if people can donate blood, we will use the plasma or the serum to generate convalescent therapy for others, and store the remaining immune cells for the donor.



