Recent years have seen incredible advances in implant technology. The most cutting-edge pacemakers today are leadless and the size of a vitamin capsule. Meanwhile, through its radical ‘electroceuticals’ initiative, British drugmaker GSK hopes to make grain-of-rice-sized bioelectronic devices that can attach to nerves and alter the signals sent between the brain and other organs a reality in the not-too-distant future.
Yet, although developments in technologies that can be implanted – and, increasingly, injected – into the body are going full steam ahead, the wireless charging systems that keep them functioning remain relatively inefficient and cumbersome.
Typically around the size of a hockey puck and worn in a gun holster-type belt, they have to line up exactly with where the implant is inside the body, meaning that the patient either needs to sit still for the two hours or more it takes their implant to fully charge, or wear it for longer than is comfortable as they move around and it continues to charge at less than optimal efficiency.
When worn for too long, the power being transferred from the charger through the skin can even result in thermal wounds.
A breakthrough technology for wireless charging
Following discussions with both the companies involved in the business of manufacturing implantable neuro-stimulators, pacemakers and the like, and the doctors dealing with the patients being treated with these devices, product design and development firm Cambridge Consultants decided to develop a solution.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
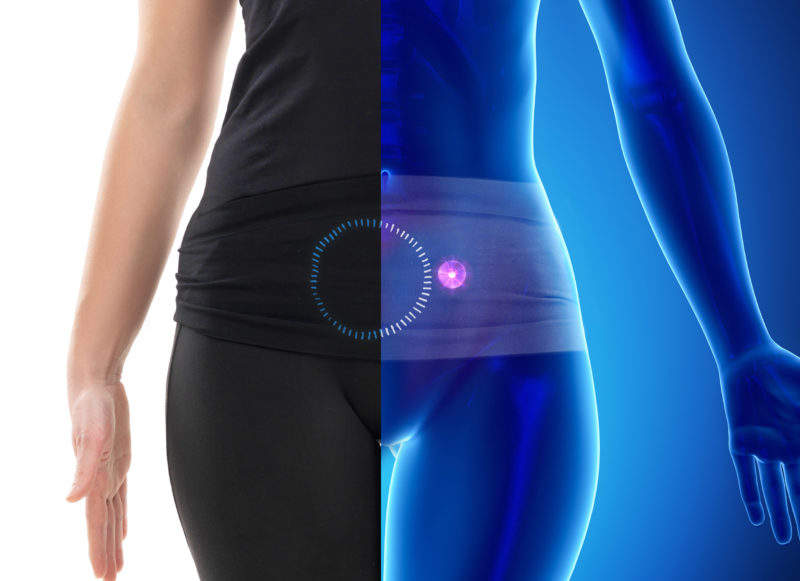
By GlobalDataA breakthrough in through-body wireless power transfer technology targeted at the manufacturers of today’s tiniest and most advanced implantable devices, the MagLense system enables flexible, efficient and safe wireless power transfer to devices inside the body without precise alignment with the implant, and regardless of the size and body shape of the patient, thanks to two new ‘novelties’.
The first is the company’s use of multiple uniquely shaped flexible coils, which can bend and flex without any impact on performance.
“There are patients with different body types and different implants go in different parts of the body – we needed to come up with a technology that was agnostic to all of that,” says Dr Arun Venkatasubramanian, head of implanted connectivity at Cambridge Consultants, and one of the two-man team behind the invention of MagLense. “If we had just come up with a technology that was flexible, there would be nothing new about that – it would be the same as existing phone charging pads. But with MagLense, when you bend it, it still maintains its performance. It’s not wasting energy in the form of heat on the skin.”
The second “clever bit of technology”, as Venkatasubramanian puts it, is the fact that MagLense is agnostic to the orientation of the implant. Newer pacemakers, which are implanted directly into the patient’s heart through minimally invasive surgery, are not only much smaller than their conventional counterparts, they can also move around within the body.
MagLense’s software is self-calibrating so that it can deliver the optimum power for different implant locations, orientations, sizes and shapes. In addition, it intelligently targets only the intended implant – avoiding any heat damage to surrounding tissue or other implants.
A new era of treatment
According to its inventor, MagLense could open up almost the whole body to the possibility of medical implants, heralding a new era of treatment for people with chronic and episodic conditions such as epilepsy, diabetes, obesity and depression. It could also enable more widespread use of micro implants for targeted nerve stimulation, the sort of innovation GSK’s electroceutical approach is designed to eventually bring to market.
“There are several new applications and we’re talking to multiple companies about how MagLense could be used to power up their next generation of bioelectronic medicines or their next generation of traditional implants,” Venkatasubramanian says.
As Cambridge Consultants is a design and development firm – the team doesn’t build and sell commercial products – the hope is that some of these conversations will lead to actual product development. The challenge now is figuring out where the technology can deliver the greatest value.
“We have created the IP, we have created the novelty aspects, we’ve shown it works,” Venkatasubramanian says. “Now we’re talking to these companies about where the biggest bang for the buck is – where the technology is absolutely needed or the system won’t work rather than being a ‘cute addition’.”
Improving patients’ quality of life
The biggest driver behind MagLense’s development was feedback from doctors about the negative impact of existing wireless charging systems on quality of life. In this regard, Venkatasubramanian is confident the new system will make a difference.
“Because of the clever algorithms and coil designs that went into MagLense, and the fact that it is agnostic to implant orientation and position in the body and automatically calibrates itself when patients move, patients are not restricted in their quality of life,” he says. “They don’t have to minimise movement – they can go for a jog and do all sorts of things and wear the belt and it will charge. That’s the whole purpose of why we started this.”
While there is certainly scope for the technology to be used for consumer devices too, Venkatasubramanian doesn’t believe this is where its greatest potential lies.
“In the consumer world, the emphasis is on usability, whereas in the medical world, it’s on performance,” he concludes. “If you have a patient with a pacemaker who has to wear a charger so his pacemaker continues to work, that’s life critical, whereas if you have a body patch that is recording your sweat or hydration levels and you have to wear a shirt with this technology in it to charge it up, it’s a different story.
“The criticality of those two scenarios and the level of performance needed for them both is very different. So while MagLense is physics, it’s technology and it could be used for either industry, its bigger value is with the medical industry.”








