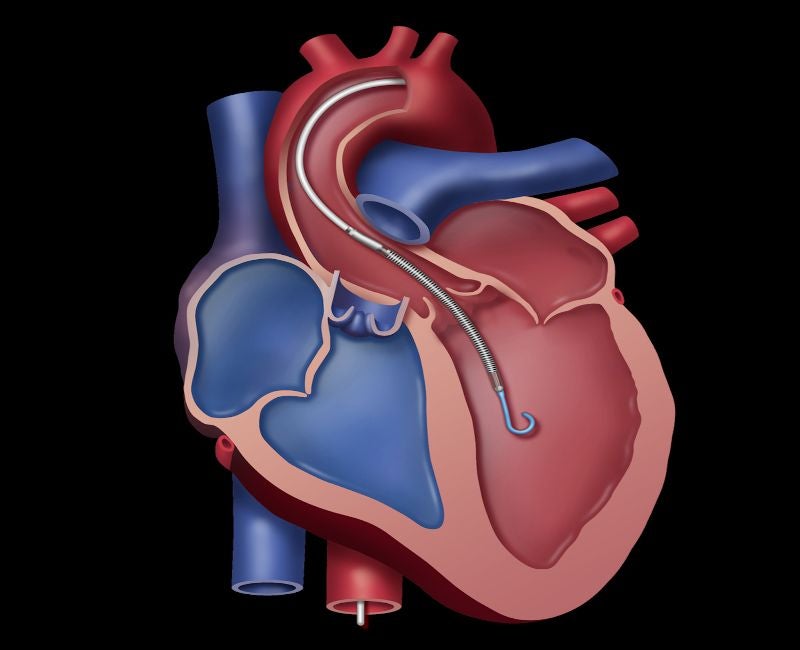
Medical devices maker Abiomed has received pre-market approval (PMA) from the US Food and Drug Administration (FDA) for its Impella 5.5 heart pump in cardiogenic shock treatment.
The minimally invasive, forward-flow device provides coronary flow and renal perfusion. It can be implanted through the axillary artery or anterior aorta, eliminating the requirement for a sternotomy or left ventricle coring.
Impella 5.5 offers full unloading to minimise the heart’s oxygen demand, said Abiomed.
Furthermore, the pump comes with SmartAssist, which features weaning algorithms to optimise survival and heart recovery. The functionality also has data informatics such as left ventricular pressure (LVP), end-diastolic pressure (EDP) and cardiac power output (CPO).
The SmartAssist fiberoptic pressure sensor is designed to enable accurate positioning, management and repositioning of the pump in ICU.
Moreover, Impella Connect facilitates the view of the Impella control screen through a website, for tracking and reviewing cases.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataFDA approved Impella 5.5 as a temporary ventricular support device for short-term use up to 14 days.
The device will provide treatment for ongoing cardiogenic shock, which occurs within 48 hours after an acute myocardial infarction, open heart surgery or cardiomyopathy.
Patients with cardiogenic shock associated with myocarditis from isolated left ventricular failure, not responding to optimal medical management and standard treatment may also benefit from the device.
According to the company, Impella System Therapy is meant to minimise ventricular work and offer circulatory support for heart recovery and early evaluation of residual myocardial function.
The company is planning a controlled launch of the Impella 5.5 with SmartAssist, which also holds the European CE-Mark, at select hospitals in the US.
Abiomed’s portfolio includes Impella heart pumps designed to treat advanced heart failure and cardiogenic shock indications.



