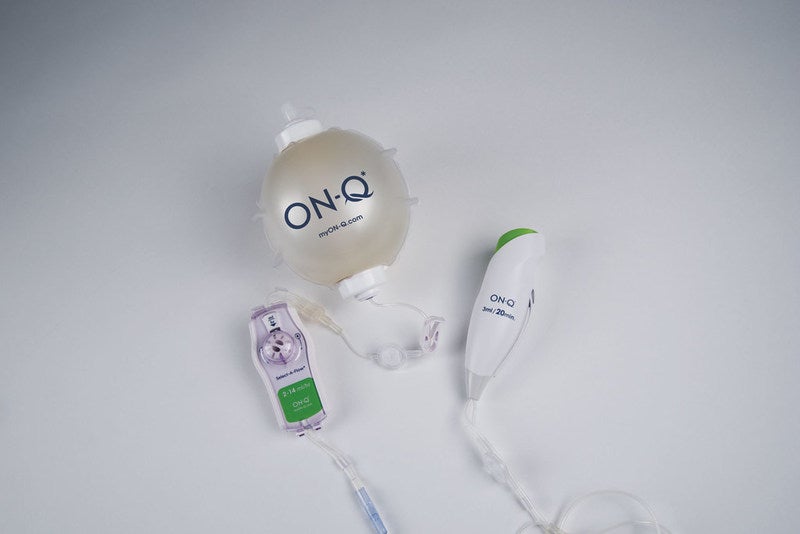
Avanos Medical has secured US Food and Drug Administration (FDA) 501(k) clearance for its postoperative pain management device ON-Q Bolus.
The device is designed to reduce the US’s reliance on opioids, offering an easy-to-use alternative for patients that are recovering from post-surgical pain.
According to studies, the administration of discrete amounts of medication (as low as 3ml) in addition to the basal rate can cut the total amount of anaesthetic required for pain relief.
ON-Q Bolus is intended to offer improved pain relief compared to opioids alone.
The device is based on the ON-Q pump, which automatically delivers a regulated flow of local anaesthetics to the surgical site or peripheral nerves. The new device offers customised control for patients for up to five days. It features an easy-to-remove priming tab and large level indicator markings.
Avanos Medical CEO Joe Woody said: “The changes we have made to the ON-Q Bolus pump are a direct result of the feedback we heard from our customers.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalData“Non-opioid approaches to pain management continue to be an important issue in the healthcare industry, particularly in light of the opioid crisis.
“As a leader in non-opioid treatment for acute pain, we are committed to continuously improving our ON-Q pumps to deliver increased quality of life and satisfaction to all patients.”
The pump will be commercially launched in the near future.
Avanos Medical’s portfolio features other pain pumps, including the ON-Q TRAC and ON-Q T-bloc Trays.


