Becton, Dickinson and Company (BD) has received a $24m investment from the US Government to support the manufacturing scale-up of Covid-19 diagnostic tests.
The investment has been made by the US Department of Defense and the US Department of Health and Human Services.
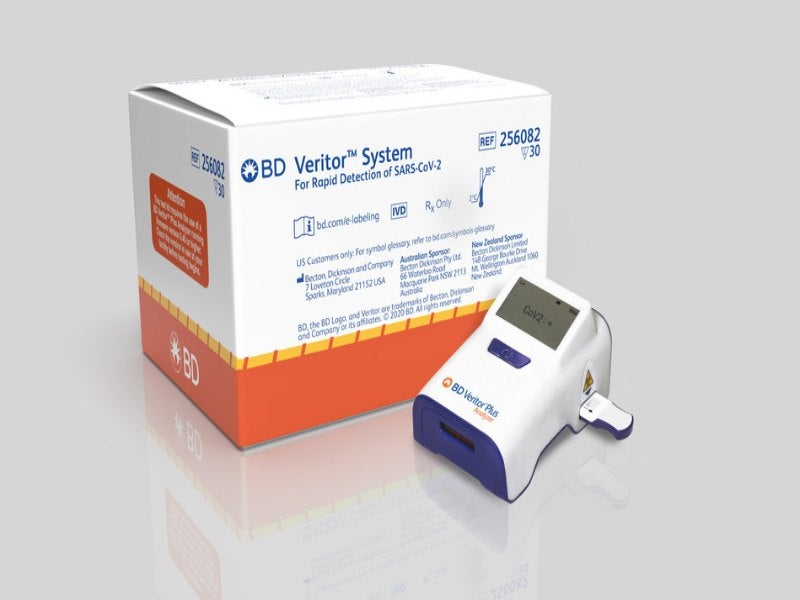
The fund will be used to expand the manufacturing capabilities of the company’s BD Veritor Solution for Rapid Detection of SARS-CoV-2.
The company plans to use the additional capital to boost domestic production and increase total production capacity by 50%.
It is estimated that the investments will ramp up global production to more than 12 million test kits a month by the end of February 2021.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataBD Integrated Diagnostic Solutions president Dave Hickey said: “Making Covid-19 diagnostic tests widely available is critical to expanding rapid detection of Covid-19 infections and mitigating the impact of the disease by identifying affected patients, quickly quarantining infectious individuals and tracing their contacts.
“This investment will bolster our US manufacturing capabilities, helping us quickly scale our production of point-of-care Covid-19 tests to ensure we have a robust supply for our US customers.”
BD obtained the Food and Drug Administration (FDA) emergency use authorisation (EUA) for the BD Veritor Plus SARS-CoV-2 antigen assay earlier this month. The assay was developed to be used in healthcare settings for the rapid diagnosis of Covid-19 in symptomatic individuals.
The company plans to leverage the US-installed base of over 25,000 BD Veritor Plus instruments to fuel the deployment of the SARS-CoV-2 assay across the country.
Earlier this month, BD formed a strategic partnership with the Biomedical Advanced Research and Development Authority (BARDA) in the US to establish new manufacturing lines for injection devices.
The company recently secured additional orders from the US and Canada governments for 177 million syringes and needles to support Covid-19 vaccination efforts.