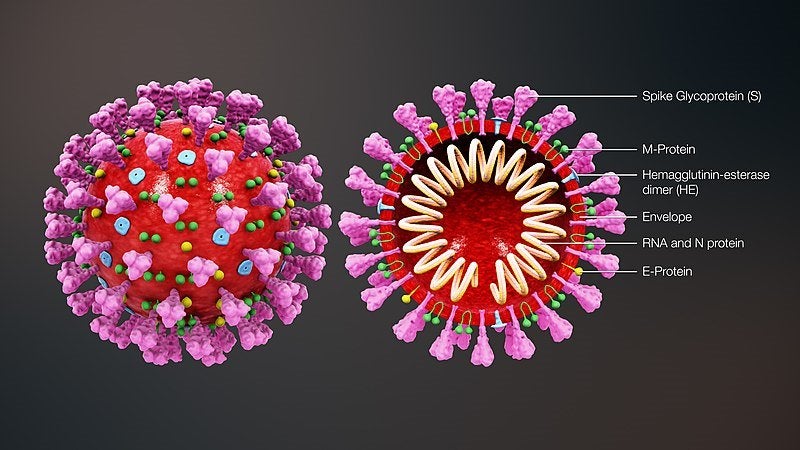
Visit our Covid-19 microsite for the latest coronavirus news, analysis and updates
Follow the latest updates of the outbreak on our timeline.
US-based firm Cepheid has announced it is developing an automated molecular test to detect the new coronavirus strain (2019-nCoV).
The company moved to develop the test as a response to the recent outbreak in China. As of 10 February, the epidemic killed more than 1,000 people with global confirmed cases surging to around 43,000.
Cepheid test is for use on its GeneXpert Systems, expected to deliver point-of-care results in around 30 minutes. According to the company, they deploy 23,000 GeneXpert Systems worldwide.
Once finalised, Cepheid plans to seek Emergency Use Authorization from the US Food and Drug Administration, delivering the assay worldwide.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataCepheid chief medical and technology officer Dr David Persing said: “The emergence of the 2019-nCoV outbreak has put significant pressure on healthcare facilities.
“An accurate test with a rapid turnaround time delivered close to the patient will expedite diagnostic testing and help alleviate these pressures.
“We plan to leverage the design principles of our current Xpert Xpress Flu / RSV cartridge technology, in which multiple regions of the viral genome are targeted to provide rapid detection of current and future pandemic coronavirus strains.”
In another development, Finnish company Mobidiag also initiated the development of Novodiag molecular diagnostic test. The test will work to identify new coronavirus 2019-nCoV, as well as other influenza viruses in 30 minutes.