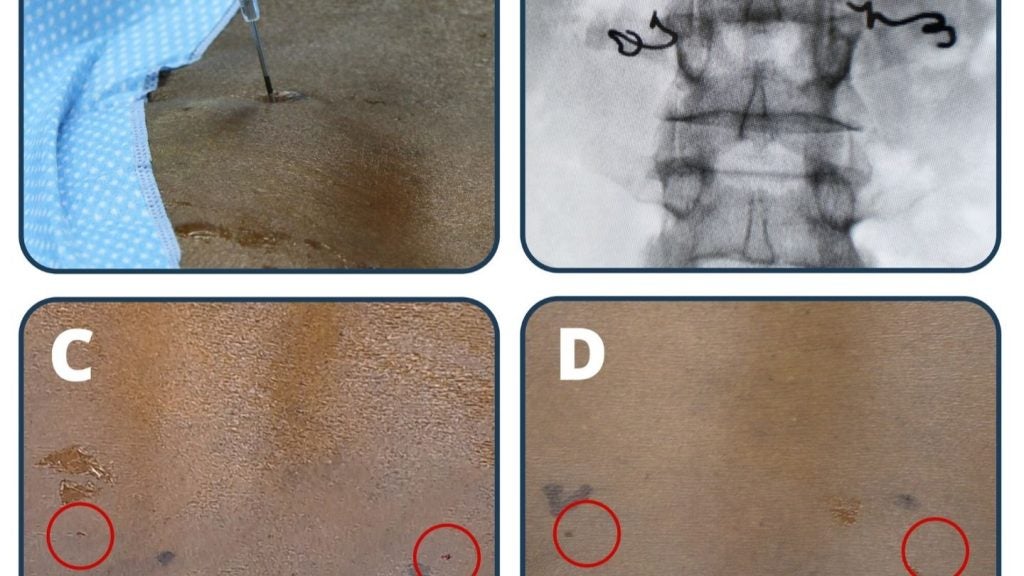
Scientists from the University of British Columbia have used electrical implants to alleviate the ‘invisible yet debilitating’ side effects for a man suffering from a spinal cord injury.
Canadian Isaac Darrel opted for the experimental treatment – which involves the surgical implantation of electrodes over his spinal cord – in 2016 following injuries sustained in a diving accident in 2012. A report on his experiences published today in JAMA Neurology said he experienced ‘dizziness, fluctuations in blood pressure and changes in bowel function’.
“Mobility issues or paralysis are the most visible consequences of a spinal cord injury but as a clinician, I know that many of my patients suffer from other ‘invisible’ consequences,” said Dr Andrei Krassioukov, principal investigator of the study and professor of medicine at the University of British Columbia, who has worked with Darrel over a number of years.
“This is one of the aspects of which I am very vocal but for many patients, as you know, not a lot of people like to talk about how we’re pooping, how we pee,” he explained to me. “And that has recently become a problem where clinicians don’t ask these questions, many patients will be shy sometimes to share these issues, and that’s why in the last, I believe, ten years, we as a research and clinical community and as well as with pressure from our patients, we start to talk more and more about invisible issues.”
The implanted electrodes are operated by Darrel using a remote control. When they are activated – as they can be for up to 45 minutes each day – they stimulate the nerve cells in the spinal cord with electrical currents. These currents mimic the signals that would come from the brain, and are designed to stimulate specific nerves that help with various motor functions. This type of treatment has long been used to alleviate chronic back pain, but only exists as a treatment for spinal cord injuries in experimental form.
These symptoms are part of a disorder known as orthostatic hypotension, which results from poor cardiovascular function. A team from the International Collaboration On Repair Discoveries (ICORD) in Vancouver used a device called a tilt table to place Darrel in an upright position to measure his blood pressure once he had undergone surgery.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalData“I’ve had better blood pressure, better core muscle, much improved bowel function, and basically I have more energy,” said Darrel, commenting that he can now sit in his wheelchair for up to eight hours, a significant improvement from the two hours he could endure before his operation.
The team behind the treatment have begun to collaborate with colleagues in the US on a larger trial to examine the benefits of the technology on a larger group. They are also working on a similar and non-invasive version of the treatment, involving stimulation through a device placed on the top of the skin rather than surgically inserted, at the University of California, Los Angeles.
Krassioukov, who is part of the International Standards Committee of the American Spinal Injury Association, considers these collaborative efforts to be integral to the success of the technology. “I know that there are numerous clinical trials that are presently approved by the FDA for examination of effects of epidural stimulation on recovery of motor functions in individuals with spinal cord injuries. To my knowledge one of the largest trials is presently run in Minneapolis, clinical trials run by Dr David Darrow, a neurosurgeon,” he told me. “My team is collaborating with this centre and we also included numerous clinical outcomes in order to examine effects of this intervention on crucial bit invisible consequences of spinal cord injuries.”
“We see very interesting and exciting results but as a clinician-scientist, I need more robust data before I would recommend this procedure,” said Krassioukov. “But I can tell you, more papers are coming!”