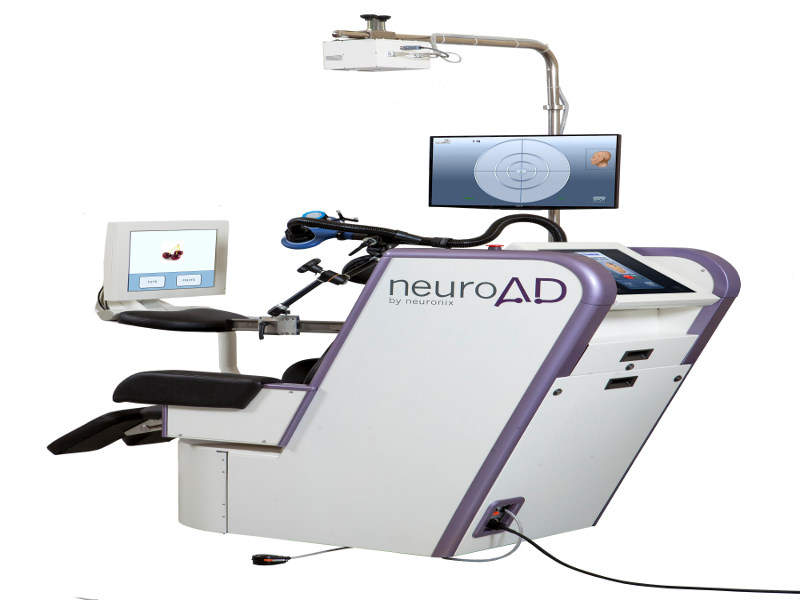

Israel-based medical device company Neuronix has reported positive results from its clinical study of the neuroAD Therapy System to treat mild to moderate Alzheimer's disease.
The neuroAD system is a non-invasive medical device based on patented non-invasive cortical enhancer (NICE) technology, which uses a combination of transcranial magnetic stimulation (TMS) with cognitive training (TMS).
TMS is used to stimulate targeted areas in the brain considered responsible for cognitive functions which have been affected by Alzheimer’s disease.
The stimulation triggers long-term potentiation (LTP) which is responsible for learning and memory processes, and makes the areas receptive to cognitive training. Such areas are subjected to tailored cognitive training and are magnetically stimulated.
The pivotal double-blind, placebo-controlled, multi-centre clinical study enrolled 131 patients to test safety and efficacy of the neuroAD Therapy System compared with placebo.
See Also:
The comparison was determined on the basis of the cognitive and behavioral standard scales for the evaluation of patients with Alzheimer's disease which are ADAS-Cog and CGI-C, respectively.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataThe therapy system demonstrated positive efficacy result for patients with milder cases of the disease which was determined on the basis of the Baseline ADAS-Cog.
After 12 weeks of treatment, the entire patient population experienced improvement, based on CGI-C results.
Additionally, the neuroAD system has demonstrated safe administration with seizures or other persistent, serious adverse events observed among the patients.
Barrow Neurological Institute Alzheimer's and Cognitive Disorders Programme director Dr. Marwan Sabbagh said: "The Study shows that treatment with the neuroAD Therapy System shows very mild and transient side effects, and can be administered without interruption to on-going pharmacological interventions.
“It was clearly demonstrated that neuroAD has the potential to improve the patients' cognitive capabilities, as well as increase their day-to-day level of activities, especially for a clinically well-defined sub-group at the milder stages.”
The results along with other study results on a similar note supported the US Food and Drug Administration (FDA) de-novo application filed by Neuronix last year for regulatory clearance of the neuroAD Therapy System for Alzheimer's disease.
Image: Neuronix’s neuroAD Therapy System. Photo: courtesy of Neuronix.



