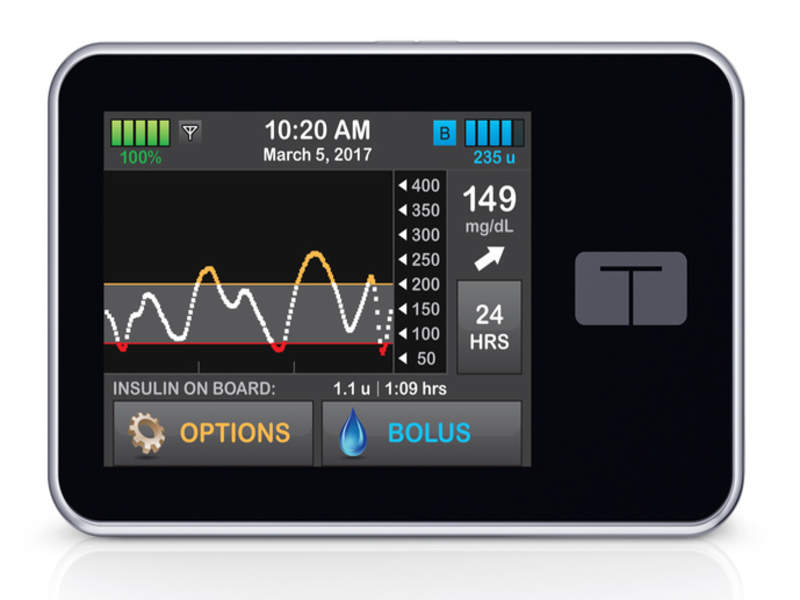

US-based Tandem Diabetes Care has started patient enrolment for the PROLOG clinical trial of its t:slim X2 Insulin Pump featuring predictive low glucose suspend (PLGS) technology.
The pump comes with an integrated Dexcom G5 Mobile Continuous Glucose Monitor (CGM) and is being developed to suspend insulin delivery upon the prediction of low blood glucose and subsequently restore insulin delivery following rising glucose levels.
Comprising two three-week treatment periods, the multi-centre, randomised, crossover PROLOG trial will compare at-home use of the t:slim X2 Pump with PLGS and a standard CGM-integrated t:slim X2 Pump without automated insulin suspension.
Tandem Diabetes Care president and CEO Kim Blickenstaff said: “The start of this pivotal trial is another important step forward in our automated insulin delivery programmes, and comes on the heels of very encouraging feasibility study data.
“We remain on track to submit our t:slim X2 Pump with predictive low glucose suspend to the FDA in early 2018.”
PROLOG is designed to enrol 90 type 1 diabetes patients aged six years and above at five US sites.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataThe trial’s primary endpoint is to demonstrate a decrease in the percentage of CGM values below 70mg/dL with the firm’s PLGS algorithm.
Tandem Diabetes Care intends to obtain approval from the US Food and Drug Administration and commercialise the t:slim X2 Insulin Pump next year.
The firm plans to provide current t:slim X2 customers with access to its new PLGS feature through a remote software update using its Tandem Device Updater.
Image: Tandem Diabetes Care t:slim X2 Insulin Pump. Photo: courtesy of Tandem Diabetes Care Inc.