US-based biotechnology firm Promega has signed a global collaboration deal with Merck to develop its microsatellite instability (MSI) technology as an on-label, solid tumour companion diagnostic (CDx) for Merck’s anti-PD1 immunotherapy pembrolizumab (Keytruda).
Initially, the companies will pursue regulatory approvals in the US and China for Promega MSI CDx, with the plan to then seek approval in other territories.
Promega president and CEO Bill Linton said: “It is gratifying to see our MSI technology have such meaning within the oncology community. Promega developed this technology well over a decade ago and our long-term commitment to R&D helped evolve its use.”
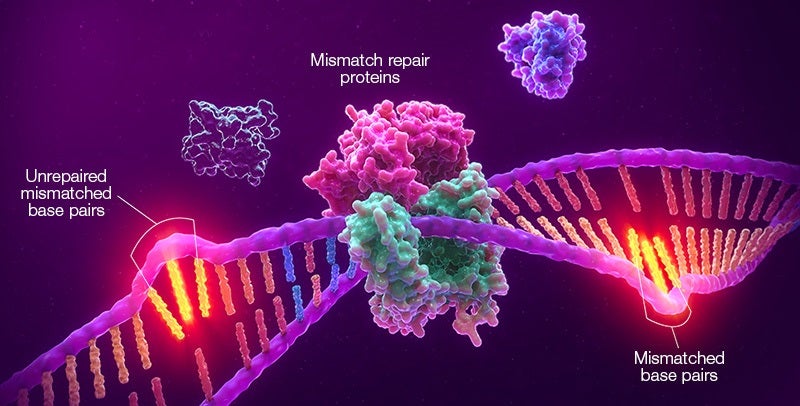
Promega’s MSI technology is a lab-validated test to characterise the MSI status of solid tumours.
The MSI test detects any genomic accumulation of insertion or deletion (INDEL) errors caused due to a deficient mismatch-repair system (dMMR), which happens in some types of solid tumours.
Tumours that have MSI-High status have indicated to respond to immune checkpoint inhibitor (ICI) therapies. This outcome may be due to MSI-driven tumour expression of mutation-associated neoantigens (MANA), considered to cause immune cell infiltration into the tumour’s microenvironment.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataIt is possible to overcome tumour-induced inhibition of immune cell activity with ICI therapies, thereby enabling tumour cell destruction by the immune cells.
Promega Corporation senior research scientist Jeff Bacher said: “Unlike other DNA-based, molecular screening options, Promega MSI technology uses five monomorphic mononucleotides, which is recommended by the National Cancer Institute.
“Our test uses a sensitive and specific panel of markers for detection of MSI status and offers valuable insight to help inform physicians on how best to treat patients with cancer including those likely to benefit from immune checkpoint inhibitor treatment.”
Promega MSI technology is one of the leading tests for MSI status detection in research laboratories, recently securing innovation status and priority review from the National Medical Products Administration (NMPA) in China.
Alongside the collaboration with Merck, known as MSD outside the US and Canada, Promega plans to seek regulatory approval for an MSI in vitro diagnostic test in the US, China and Europe, with a prospective launch of 2020 in these regions.