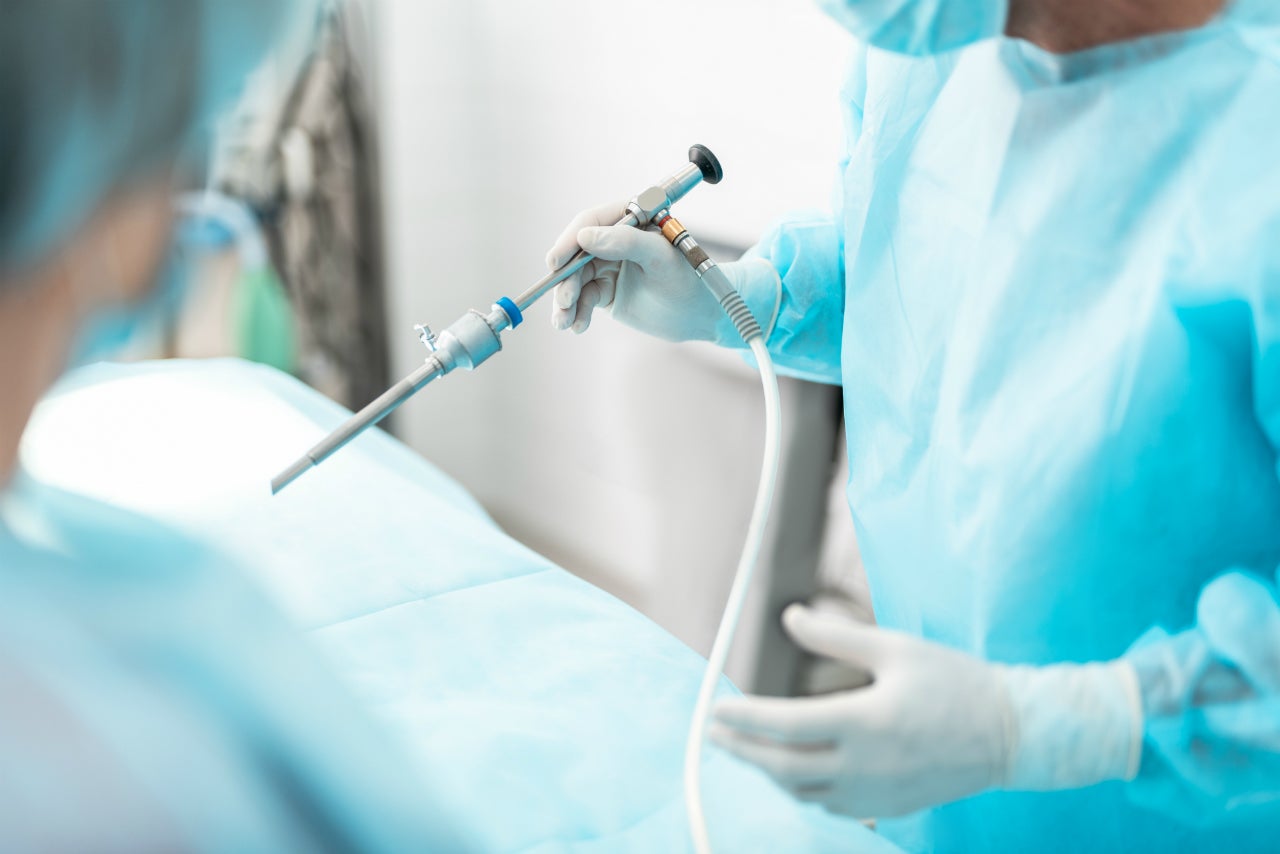
US-based medical device company Xenocor, which specialises in the development of laparoscopic devices, has announced an investment from Mountain Pacific Venture Fund (MPVF).
The company’s single-use, multi-purpose HD laparoscope, called Xenoscope, is designed to reduce common visualisation challenges and offer consistent image quality through electrocautery smoke.
Furthermore, the Food and Drug Administration (FDA)-cleared and CE-marked reduces the risk of disease transmission and operational downtime by eliminating the need to sterilise the scope pre-surgery.
MPVF’s investment will support Xenocor’s commercialisation efforts for the device with an initial focus on the growing ambulatory surgery centre (ASC) market.
Xenocor CEO Evan Kelso said: “We set out to make a more effective and reliable surgical device, which would enhance the quality of care for patients, but we have also dramatically impacted the bottom line for hospitals and surgery centres by reducing upfront capital expenditures and eliminating both service contracts and maintenance costs, making it more accessible to those who need it.”
MPVF managing partner June Chen said: “We are seeing a steady increase in demand for single-use scopes, driven by a rise in hospital-acquired infections and a push from the FDA to start transitioning away from devices that require reprocessing.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalData“These factors, along with the current pandemic, are fueling a sea of change where more procedures will be done outside of the hospital in lower-risk settings such as outpatient clinics or ambulatory surgery centres where Xenoscope can really have an impact.”
Separately, Xenocor has signed a group purchasing agreement with healthcare improvement company Premier for the Xenoscope in the surgical disposable scope category.
According to the three-year agreement, Premier members will benefit from pre-negotiated special pricing and terms in connection to Xenocor’s laparoscopic device.
The agreement will be effective from the beginning of next month.





