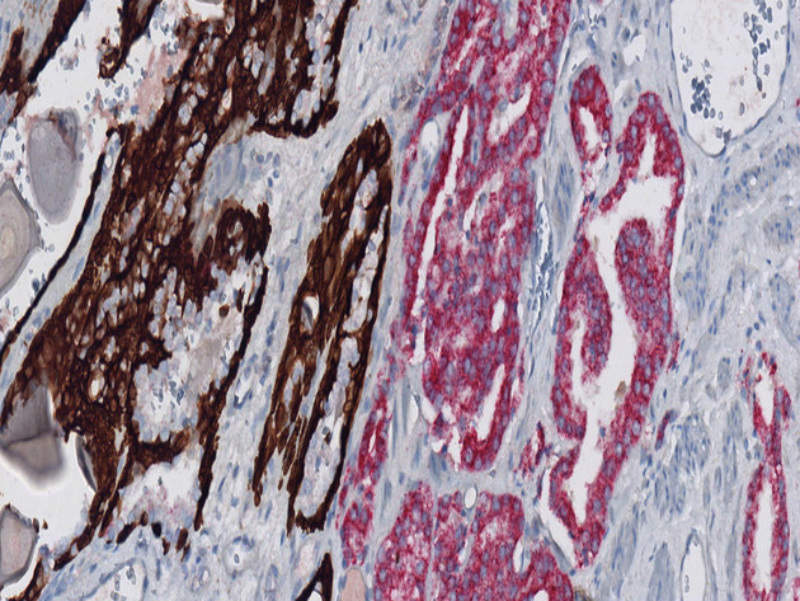

Roche launches new prostate cancer diagnostic test
Swiss diagnostic firm Roche launched a new test called anti-p504s (SP116) Rabbit Monoclonal Primary Antibody for the diagnosis of prostate cancer.
Intended for the use in laboratories, the test is designed to detect α-methylacyl-CoA racemase (AMACR) in formalin-fixed, paraffin embedded (FFPE) tissue sections.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The new tool is validated for use with a dual stain and VENTANA Basal Cell Cocktail (34ßE12+p63) to enable easy differentiation of morphologically difficult cases such as benign, atypical or cancerous on a single slide.
Cardinal Health to gain Medtronic's medical products business for $6.1bn
Cardinal Health signed a definitive $6.1bn agreement to acquire Medtronic's Minimally Invasive Therapies Group’s (MITG) patient care, deep vein thrombosis, and nutritional insufficiency businesses, which are part of the group's patient monitoring and recovery (PMR) division.
In addition to $6.1bn, the agreement includes after-tax proceeds of around $5.5bn. From this, Medtronic plans to use $1bn for repurchasing incremental shares in the next financial year and the remainder going towards reducing debt.
The acquisition includes Medtronic's dental/animal health, chart paper, wound care, incontinence, electrodes, SharpSafety and thermometry. It also includes perinatal protection, blood collection, compression and enteral feeding product lines, alongside 17 associated manufacturing facilities.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataBecton Dickinson agrees to buy Bard for $24bn

US-based Becton Dickinson (BD) entered a definitive agreement to buy CR Bard for $24bn.
The firms will create a combined company to deliver healthcare solutions to both patients and care providers.
The acquisition will leverage BD's experience in medication management and infection prevention, while Bard's portfolio of products will bolster BD's opportunities in clinical areas.
Boston Scientific to purchase Symetis for $435m
US-based medical solutions provider Boston Scientific entered a definitive agreement to purchase Switzerland-based medical technology company Symetis for $435m.
The acquisition is expected to aid Symetis in expanding its treatment offerings for aortic valvular heart disease patients.
Symetis' portfolio includes the valve systems ACURATE TA and ACURATE neo/TF developed for the treatment of high-risk patients with severe and symptomatic aortic valve stenosis.
B Braun and Philips form strategic alliance to enhance regional anaesthesia and vascular access

Germany-based medical device firm B Braun entered a multi-year strategic alliance with Dutch health technology company Royal Philips to develop ultrasound-guided regional anaesthesia and vascular access.
The firms intend to combine clinical expertise, sales, service channels and R&D capabilities to jointly develop and commercialise regional anaesthesia solutions.
The solutions are expected to increase needle visualisation and guidance, as well as optimise procedure workflows and resource planning.
NYU researchers investigate new blood tests to detect skin cancers
Researchers at New York University (NYU) Langone Medical Center and its Perlmutter Cancer Center are investigating two new blood tests to detect skin cancers.
Results from genetic testing of tumour and blood fluid samples from patients with and without metastatic melanoma showed that the tests can reliably identify previously unidentifiable forms of the disease.
It is expected that the precise and quick monitoring tools will enable easy and early identification of cancer recurrence.
ResMed develops new compact device for sleep apnoea

ResMed developed the new AirMini continuous positive airway pressure (CPAP) machine for patients with sleep apnoea.
Designed to provide ease while travelling, the portable, pocket-sized device weighs 300g and measures 13.6cm x 8.4cm x 5.2cm.
AirMini features the firm's CPAP technology, AutoSet functions and the new built-in HumidX humidification system, which captures exhaled heat and moisture in the mask tube and redelivers it to the patient.
Chembio and FIND to develop POC test for multiple febrile illnesses in Asia Pacific
US-based diagnostic tests provider Chembio Diagnostics partnered with Swiss-based Foundation for Innovative New Diagnostics (FIND) to develop a point-of-care (POC) test, DPP Fever Panel Assay, to detect several acute febrile illnesses in the Asia Pacific region.
The firms will be working over the coming year to develop a simple, rapid and cost-effective test using Chembio’s Dual Path Platform (DPP) technology.
The new multiplex test will be designed to simultaneously identify multiple life-threatening acute febrile diseases, contrary to the existing POC tests.
BMS and Nordic Bioscience partner to develop biomarker technology for fibrotic diseases
US-based biopharmaceutical company Bristol-Myers Squibb (BMS) entered a collaboration agreement with Danish biotech firm Nordic Bioscience for developing new biomarker technology to detect fibrotic diseases.
The technology is expected to support the diagnosis and monitoring of fibrotic diseases, including non-alcoholic steatohepatitis (NASH).
The companies will develop diagnostics and translational biomarkers for NASH assessment in preclinical models of fibrotic disorders, as well as in clinical settings.
Strand Life Sciences introduces new liquid biopsy tests for cancer
India-based bioinformatics firm Strand Life Sciences introduced a new line of liquid biopsy tests STRAND LB to detect traces of cancer from a blood sample.
The highly sensitive test is designed for early and accurate identification of the presence of a tumour, recurrence of the disease and response to therapy.
The test claims to enable early evaluation with low patient discomfort, contrary to the invasive tumour biopsies and radioactive scans.



