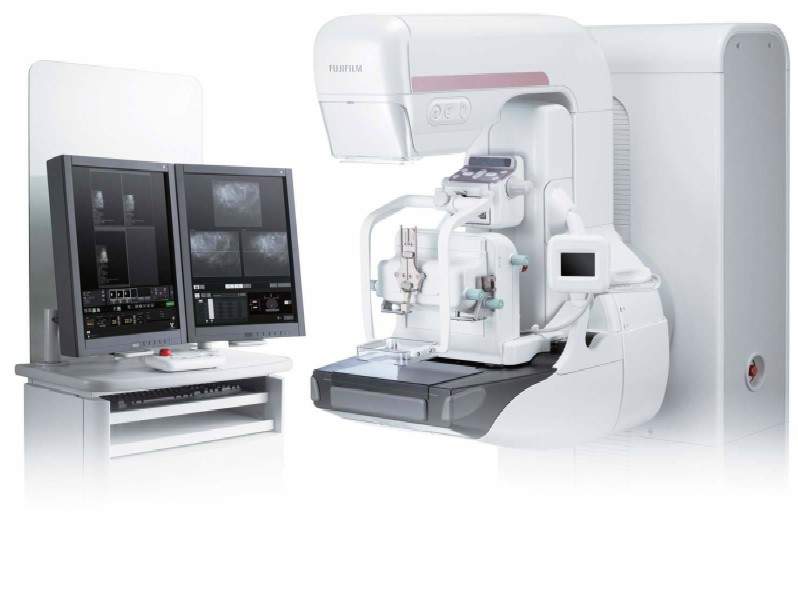

Fujifilm gets FDA premarket approval for Digital Breast Tomosynthesis
Fujifilm Medical Systems USA secured premarket approval (PMA) from the US Food and Drug Administration (FDA) for its Digital Breast Tomosynthesis (DBT) option of ASPIRE Cristalle system.
DBT has been developed as an optional software upgrade for the digital mammography ASPIRE Cristalle FFDM system.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Designed to optimise patient dose and improve image quality, the ASPIRE Cristalle FFDM system with DBT utilises a combination of hexagonal close pattern (HCP) detector design, advanced image processing and image acquisition workflow.
VolitionRx gets CE mark for Nu.Q Colorectal Cancer Screening Triage Test
Life sciences company VolitionRx secured CE mark for its Nu.Q Colorectal Cancer Screening Triage Test paving the way for its launch across the European Union (EU).
Developed in collaboration with the Hvidovre Hospital of University of Copenhagen, Nu.Q detects nucleosomic markers of cancer for an early stage detection using a series of convenient, inexpensive and accurate blood tests. The test analyses the unique nucleosomes to ascertain the presence of the disease.
The introduction of the Nu.Q colorectal cancer screening triage test will complement the existing standard screening test, the fecal immunochemical test (FIT) widely used in colorectal cancer screening programmes.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataFDA clears Luminex’s ARIES GBS assay for GBS colonisation detection in pregnant women
The US Food and Drug Administration (FDA) granted clearance for medical technology firm Luminex’s ARIES Group B Streptococcus (GBS) assay to detect GBS colonisation in pregnant women.
Also known as Streptococcus agalactiae, GBS forms colonies in one in four pregnant women, and if left untreated, can be transmitted to a newborn during labour and delivery, potentially resulting in septicemia, pneumonia or meningitis for both the mother and the newborn.
The ARIES GBS Assay is a real-time polymerase chain reaction (PCR) based qualitative in vitro diagnostic test which sensitively and accurately detects Group B Streptococcus nucleic acid from 18-24 hour Lim broth enriched vaginal-rectal swab specimens derived from pregnant women.
Abbott launches EnSite Precision cardiac mapping system in US

Abbott announced the US launch and first commercial uses of the new EnSite Precision cardiac mapping system and Advisor FL Circular Mapping Catheter, Sensor Enabled to map cardiac arrhythmias during ablation treatment.
The first commercial use of the system following US Food and Drug Administration approval occurred at Intermountain Heart Institute at Intermountain Medical Center in Salt Lake City during ablation procedures conducted by electrophysiologist Dr John Day.
Abbott expects its new cardiac mapping technology will quickly become a new standard for use in treating patients experiencing arrhythmias from multiple origins in the heart.
Hill-Rom agrees to buy Mortara Instrument for $330m
US-based medical technology company Hill-Rom agreed to acquire medical devices and technology company Mortara Instrument for $330m in cash.
The portfolio of Mortara consists of diagnostic cardiology devices for a comprehensive usage across the clinical care, from acute care to primary care and clinical research organisations.
Mortara's range of products operates under three brands which are Mortara, Quinton and Burdick, including resting electrocardiography (ECG), cardiac stress exercise, Holter monitoring, ambulatory blood pressure monitoring, and cardiac and pulmonary rehabilitation and multi-parameter patient monitoring.
Linde Gas acquires The Service Center
Linde Gas North America, a unit of Linde Group, acquired the assets and business of The Service Center, a provider of medical oxygen and respiratory equipment repair services to healthcare providers in the US.
Financial details of the transaction have not been disclosed.
Founded in US in 1997, The Service Center works with home medical equipment companies, hospitals and nursing homes and primarily offers services including medical oxygen transfilling, cylinder requalification and repair, and respiratory equipment repair, to healthcare providers in the Midwest and Southeast regions of the US.
Augmenix reports positive report from Phase III trial of SpaceOAR System

US-based medical technology company Augmenix reported positive long-term data of its Phase III clinical trial of the SpaceOAR System to separate the rectum and prostate during prostate cancer radiotherapy.
SpaceOAR System has been developed to temporarily separate the anterior rectal wall from the prostate during radiotherapy, limiting the exposure of the anterior rectum to radiation.
Radiation therapy for prostate cancer poses a risk for the adjacent healthy tissue which might cause a range of bowel, urinary symptoms that can affect patient health and quality of life during radiotherapy, and in the future.
New study suggests SCS therapy could reduce or stabilise use of opioid
New research suggests that spinal cord stimulation (SCS) therapy the potential to reduce or stabilise the use of opioids in patients battling chronic pain.
The research was sponsored by Abbott, a global company that develops and manufactures SCS systems and therapy options.
Researchers studied the opioid usage data from nearly 5,400 patients both prior to and after receiving an SCS system implant.
Mobidiag receives CE-IVD mark for Amplidiag Easy diagnostic device
Finland-based molecular diagnostics firm Mobidiag received CE-IVD marking for a proprietary system called Amplidiag Easy, which enables nucleic acid extraction and PCR setup directly from stool samples.
The device enables quicker sample to result time through an automated solution, which runs directly from stool primary sample tubes to PCR setup, eliminating the requirement for manual setup.
The system is equipped with the Amplidiag Analyzer software, which enables direct transfer of data between the Amplidiag Easy and PCR instruments.
Paragon 28 introduces new small bone fixation system for foot and ankle

US-based surgical devices company Paragon 28 introduced a new small bone fixation system for foot and ankle injuries named the Baby Gorilla Mini Plating System.
The Baby Gorilla Mini Plating System combines the modularity, optionality and specificity of the classic Gorilla Plating system.
The new system provides a set of implants for procedures that may require narrower plates and smaller screws which are similar in size and anatomy of small bones within the foot and ankle.





