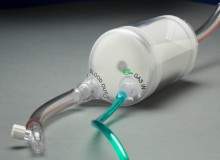

FDA grants 510(k) clearance for CardiacAssist's new TandemLung oxygenator
The US Food and Drug Administration (FDA) granted 510(k) clearance for CardiacAssist's new medical device, the TandemLung oxygenator, which acts as an artificial lung to infuse oxygen and remove carbon dioxide from the blood.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The device uses a patent-protected and optimised radial flow design, coupled with advanced polymethylpentene (PMP) fibres to transfer oxygen into the blood for patients requiring cardiac or respiratory support.
The TandemLung oxygenator is intended for use in adult patients for extracorporeal circulation during cardiopulmonary bypass for up to six hours.
St Jude's Nanostim leadless pacemaker gets CE mark for MR labelling

St Jude Medical received the conformité européenne (CE) mark approval for magnetic resonance (MR) conditional labelling for 1.5T scans of its Nanostim leadless pacemaker.
The approval allows existing and new patients implanted with a Nanostim leadless pacemaker to safely undergo full-body MRI diagnostic scans in the future.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataThe company noted that the latest approval builds upon its portfolio of MRI-compatible products, and furthers patient access to the technological benefits of the Nanostim leadless pacemaker.
Origin Technologies makes $1.5bn offer to buy Affymetrix
Origin Technologies made an offer of around $1.5bn to acquire US-based medical technology firm Affymetrix, topping a $1.3bn bid from Thermo Fisher Scientific made this January.
Origin is owned by a group of former Affymetrix executives, who intend to accelerate genomics for precision medicine and other critical applications.
The company will pay $16.10 per share to acquire Affymetrix, representing a 75% premium to its closing share price of $9.21 on the last trading day, before the announcement of Thermo Fisher Scientific.
FDA grants 510(k) clearance for Stryker's Tritanium PL posterior lumbar cage

The US Food and Drug Administration (FDA) granted 510(k) clearance for medical technology firm Stryker's intervertebral body fusion device, the Tritanium PL posterior lumbar cage.
The new 3D printed cage has been developed to help in lumbar spinal fixation of patients with degenerative disc disease, grade I spondylolisthesis, and degenerative scoliosis.
Developed using a 3D additive manufacturing process, Tritanium is a porous titanium material designed for bone in-growth and biologic fixation in spine applications.
Canon agrees to buy Toshiba Medical Systems for $5.9bn
Japan-based Canon entered into an agreement with Toshiba to acquire Toshiba Medical Systems (TMSC) for around JPY665.5bn ($5.94bn).
The companies signed a share transfer agreement to make TMSC a subsidiary of Canon.
Canon secured exclusive negotiation rights to acquire TMSC as part of Toshiba's aim to restructure its operations following an accounting scandal.
Smith & Nephew unveils first 3D-printed titanium hip implant

British medical technology firm Smith & Nephew unveiled its first 3D-printed titanium hip implant at the American Academy of Orthopaedic Surgeons (AAOS) annual meeting in Orlando, US.
At the event, the company exhibited the new Redapt revision acetabular fully porous cup with Conceloc technology.
Last November, the company received 510(k) clearance for the Redapt cup from the US Food and Drug Administration (FDA).
Roche launches Discovery Yellow kit for cancer research
Swiss global healthcare firm Roche expanded its Ventana modular-based detection kits with the launch of the new Discovery Yellow Kit in order to carry out cancer research.
The kits will support in the identification and profiling of cancer biomarkers and tumour cell populations.
Discovery Yellow is an alkaline phosphatase-based detection chemistry for tissue diagnostics, which produces a transparent yellow signal that cannot be washed during alcohol dehydration, such as some precipitating dyes, and fits into routine laboratory workflow.
Boston Scientific introduces Axios stent and delivery system

US-based medical device firm Boston Scientific launched Axios stent and electrocautery enhanced delivery systems to manage two severe complications from pancreatitis via a minimally invasive endoscopic approach.
The Axios system will assist physicians in endoscopic management of pancreatic pseudocysts and several types of walled-off pancreatic necrosis.
The conditions interpret two types of pancreatic fluid collections (PFCs), which take place in 5% to 16% of patients with acute pancreatitis, and 20% to 40% of patients with chronic pancreatitis.
J&J subsidiary Ethicon to acquire NeuWave Medical
Ethicon, a medical-device subsidiary of Johnson & Johnson (J&J), entered an agreement to acquire US-based NeuWave Medical for an undisclosed amount.
NeuWave produces and markets minimally invasive soft-tissue microwave ablation systems, which are currently used by physicians in cancer centres across the country.
The acquisition is part of a J&J strategy to invest in areas of unmet medical needs, such as surgical oncology.





