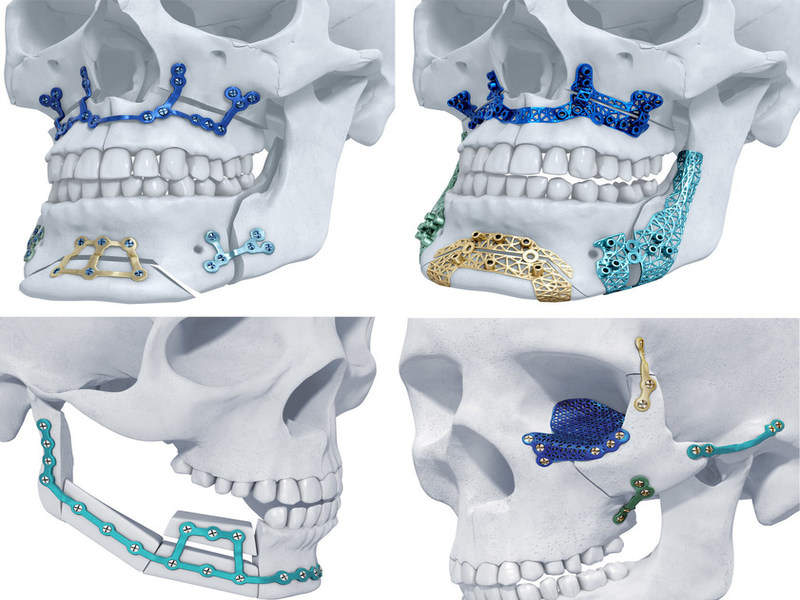

Belgium-based medical three-dimensional (3D) printing applications provider Materialise Medical has received clearance from the US Food and Drug Administration (FDA) to market its titanium implants for maxillofacial surgery.
Johnson and Johnson Medical Devices company DePuy Synthes will distribute the implants in the country.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The TRUMATCH 3D-printed implants are designed based on a CT scan of a patient’s skull to serve as a personalised solution for corrective jaw (orthognathic) surgery and facial reconstruction.
To be used with 3D-printed surgical guides, the implants are said to integrate virtual surgical planning and intraoperative patient-specific tools to aid better aesthetic results with minimal surgery time.
Materialise Medical vice-president Brigitte de Vet said: “As the first of our extensive selection of implants to receive clearance for the US markets, the decision is a real milestone for our medical department.
“Thanks to our partnership with DePuy Synthes, our devices will be able to provide better healthcare for as many patients as possible.”

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataMaterialise and DePuy Synthes entered an agreement last July to deliver the 3D-printed titanium implants for use in facial and skull disorders in Europe (except France) and Australia.
The firms have been working together since 2010 to provide personalised solutions for craniofacial surgery and help surgeons to achieve accuracy, as well as improved patient outcomes.
Materialise Medical portfolio includes virtual planning software tools, 3D-printed anatomical models, and patient-specific surgical guides and implants.
DePuy Synthes focuses on joint reconstruction, trauma, craniomaxillofacial, spinal surgery, and sports medicine.
Image: TRUMATCH Titanium 3D-printed implants. Photo: courtesy of DePuy Synthes.





