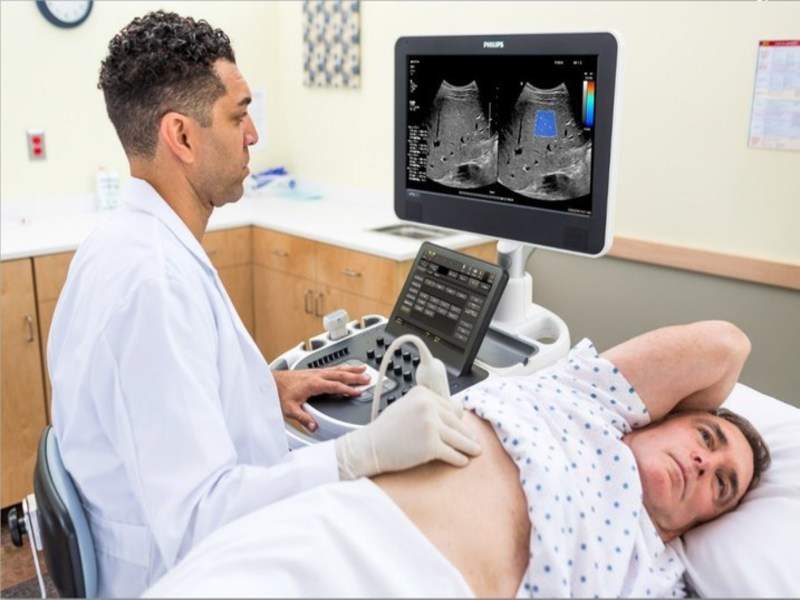

The US Food and Drug Administration (FDA) has granted 510(k) clearance to Royal Philips' new ultrasound system ElastQ Imaging.
The new system allows diagnosis of various liver conditions through simultaneous imaging of tissue and examination of its stiffness.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
ElastQ Imaging is a non-invasive, reproducible capability that utilises shear wave elastography to focus sound waves for the assessment of soft tissue stiffness.
According to a research, ultrasound exams using shear wave elastography may aid in avoiding the costly and painful liver biopsy procedures.
Philips ultrasound business leader Vitor Rocha said: "We know that liver disease is a growing health concern around the globe, and we are committed to pioneering innovations like ElastQ Imaging to create our ultimate ultrasound liver solution that offers exceptional clinical performance, further improving patient care."
ElastQ Imaging features large field of view or region of interest (ROI), colour-coded quantitative assessment of tissue stiffness, real-time feedback, intelligent analysis and quantitative measurements with multiple sample points.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataPhilips offers a non-invasive ultrasound liver solution that combines ultrasound imaging exam of the liver anatomy with targeted tissue stiffness values.
The ultrasound liver solution comprises of PureWave transducer technology, Contrast enhanced ultrasound and Image fusion and navigation with anatomical intelligence, in addition to ElastQ Imaging.
While, PureWave facilitates enhanced image quality and penetration, Contrast enables liver lesion detection, characterisation and Image fusion allows real-time ultrasound exams via multimodality fusion and interventional guidance.
Image: A doctor using the Philips' ultimate ultrasound liver solution. Photo: courtesy of Philips.





