
US-based medical device developer HeartSciences has launched its MyoVista high-sensitivity electrocardiograph (hsECG) Testing Device in Europe after receiving CE-Mark.
MyoVista is intended to provide an effective, low-cost, front-line screening solution for cardiac disease in symptomatic as well as asymptomatic patients.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
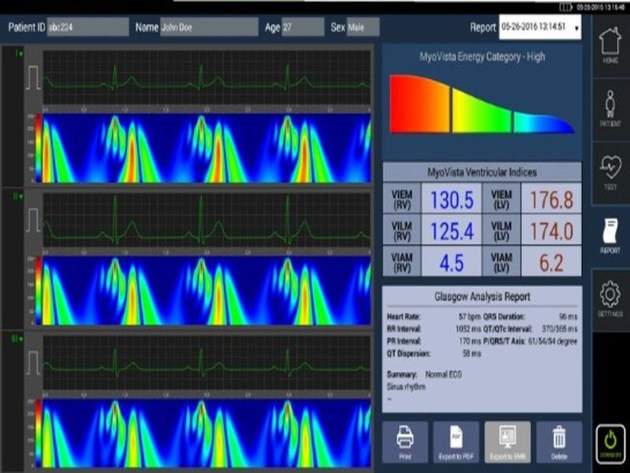
The device is designed to allow early detection of the disease by measuring the heart's energy during each heartbeat with an advanced signal processing technology called Continuous Wavelet Transform (CWT).
It delivers a detailed visual image of the energy distribution that occurs during a cardiac cycle.
HeartSciences chairman Andrew Simpson said: "Currently, there's a significant diagnostic gap in detecting heart disease early, resulting in a burden on both patients and healthcare systems.
"We believe MyoVista hsECG could play an important role in achieving the preventative treatment ambitions of many healthcare systems, as well as help reduce unnecessary healthcare expenditures."

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataIn a validation trial conducted to identify left ventricular diastolic dysfunction in 200 subjects, the MyoVista hsECG technology demonstrated 88% sensitivity and 87% specificity for the detection of cardiac dysfunction in the target cardiac cycle.
MyoVista employs a 12-lead, at-rest testing protocol that is similar to the existing ECG devices.
The device provides resting ECG tracings, ECG interpretive analysis and additional informatics to enable detection of cardiac dysfunction associated with coronary artery disease (CAD) and structural disease, as well as arrhythmias.
HeartSciences intends to expand distribution of MyoVista to Australia, the Middle East, Latin America, Asia-Pacific, and Canada, with plans to apply for the US Food and Drug Administration (FDA) clearance next year.
Image: The Testing Device provides physicians with a detailed visual image of the energy distribution during the cardiac cycle. Photo: courtesy of PRNewsfoto/HeartSciences.





