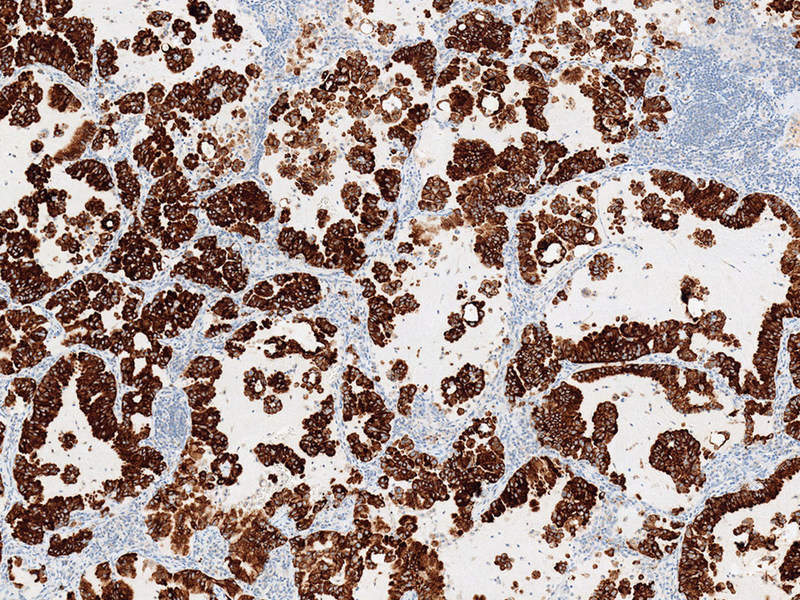

Roche has received CE-Mark for its Ventana anti-ALK (D5F3) Rabbit Monoclonal Primary Antibody as a companion diagnostic for patients with non-small cell lung cancer (NSCLC).
The immunohistochemistry (IHC) test can now be used to identify ALK-positive NSCLC patients who are eligible for treatment with the Zykadia (ceritinib) drug provided by Novartis.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Meant for laboratory use, the in-vitro diagnostic (IVD) test is designed to detect the anaplastic lymphoma kinase (ALK) protein in NSCLC tissue that is fixed with formalin, embedded in paraffin and stained using the firm’s BenchMark immunohistochemical automated slide stainers.
As an important biomarker for NSCLC, ALK’s detection and inhibition are expected to aid shrinkage of tumours in certain ALK-positive patients.
Roche Tissue Diagnostics head Ann Costello said: “Precise, personalised diagnostics are critical as we continue the fight against non-small cell lung cancer.
“Expanded use of the Ventana anti-ALK (D5F3) Rabbit Monoclonal Primary Antibody to identify non-small cell lung cancer patients with an ALK-positive mutation allows us to aid clinicians and their patients in identifying appropriate treatment options.”

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataThe Ventana anti-ALK (D5F3) Rabbit Monoclonal Primary Antibody is currently available for use on the BenchMark IHC/ISH instruments.
In addition to Zykadia, the test is indicated to identify patients eligible for treatment with Xalkori (crizotinib).
Founded in 1896, Roche develops diagnostics and pharmaceuticals for personalised healthcare in oncology, immunology, infectious diseases, ophthalmology and central nervous system disorders.
The firm focuses on IVDs, tissue-based cancer diagnostics and diabetes management.
Image: NSCLC stained with the Ventana anti-ALK (D5F3) Rabbit Monoclonal Primary Antibody and OptiView DAB Detection and Amplification. Photo: Courtesy of PRNewswire.





