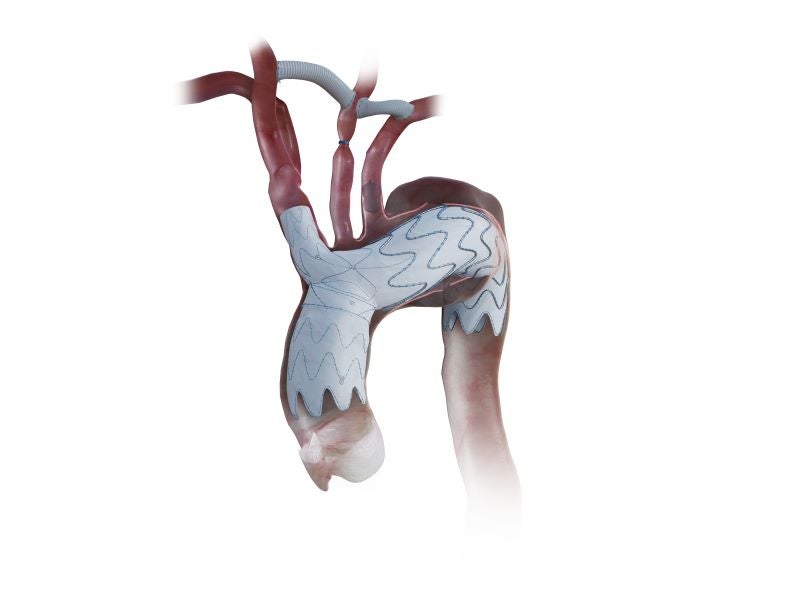
The NEXUS® aortic arch stent graft system is an off‑the‑shelf endovascular solution for treating aortic arch pathologies including chronic dissection, aortic aneurysm, intramural haematoma and penetrating aortic ulcer.
Developed by Endospan, an Artivion company, the device provides a minimally invasive option for patients considered at high risk for open surgical repair and uses a modular configuration with an integrated branch component to address disease in the aortic arch.
Thoracic aortic arch disease leads to the loss of strength in the aortic wall within the arch section of the thoracic aorta (the body’s primary artery). A compromised segment may tear or rupture, leading to major internal haemorrhage that can result in shock or be fatal. Thoracic aortic arch conditions affect more than 120,000 people annually across the US and Europe.
NEXUS is approved in Europe and is reported to be the first CE‑marked, commercially available endovascular aortic arch branch system in the region, indicated for the repair of both aneurysms and dissections in the aortic arch.
In April 2026, the system obtained premarket approval from the US Food and Drug Administration (FDA) for the treatment of aortic arch disease.
NEXUS stent graft system design and features
The NEXUS stent graft system is designed to optimise blood flow by maximising cross‑sectional area, supporting integrated flow, reducing turbulence and promoting efficient haemodynamics to lower cardiac workload.
The system is composed of three elements: an ascending aorta component, an aortic arch component and locking latches.
The ascending aorta stent graft has a distal diameter of 36mm and is available in centreline lengths of 40mm/55mm/70mm and a proximal diameter of 36mm/40mm/43mm. It incorporates a long outer curve and a short inner curve to align with the natural course of the ascending aorta, to enhance conformability, fixation and sealing.
The stent tips positioned along the outer curve of the ascending aorta are bent inward. This configuration is intended to create a less traumatic interface between the stent graft and the aortic wall.
The aortic arch component of the NEXUSsystem has a docking diameter of 30mm and is available in distal diameters of 32mm/36mm/40mm/44mm. The arch’s retrograde inner branch measures 10mm in diameter and 20mm in length, while body length is 130mm.
The inner branch incorporates dedicated sealing areas to secure the covered stent. Its reverse‑oriented configuration enables transfemoral delivery of every component while preserving access for potential future reinterventions via the supra‑aortic vessels.
The aortic arch component also incorporates an integrated branch, which is intended to accommodate a broad range of anatomical variations. The integrated branch is available in diameters of 14mm/17mm/20mm and lengths of 20mm/30mm/40mm.
A proprietary locking mechanism is designed to provide stable anatomical fixation and atraumatic sealing across varied anatomies. It uses two distal struts with locking latches to secure the inner stent graft within the docking sleeve, aiming to prevent disconnection between modules and achieve mechanical fixation. The locking mechanism establishes a 24mm sealing length with approximately 20% oversizing.
NEXUS stent graft delivery system
The NEXUSdelivery system enables the graft system to be introduced through vessels with diameters as small as 7mm. The low‑profile, 20 French, fully transfemoral delivery system is designed to reduce access-related complications and lower the risk of stroke.
The system utilises a distinct deployment sequence designed to protect the brachiocephalic artery from embolic debris that might otherwise travel to the brain.
Key elements of the delivery system include a pre‑shaped hydrophilic catheter, dual flushing ports, a distal shaft flushing port and an internal fixation tube with tether suture.
The pre-shaped catheter is engineered to track the natural curvature of the aortic arch, minimising the need for manipulation and supporting stent conformability during deployment.
The preloaded guidewires provide straightforward entry to the stent graft’s inner branch, helping to minimise instrument handling and shorten procedure time.
Both flushing ports are designed to maximise air removal from the sheath.
Clinical trials on NEXUS system
The US FDA approval of the NEXUSsystem was supported by data from the TRIOMPHE multi-centre investigational device exemption study evaluating the safety and effectiveness of the system in patients at high surgical risk.
The study enrolled 110 high‑risk surgical patients with chronic aortic dissections, focusing on treatment of the ascending aorta, a segment associated with elevated rates of complications such as mortality and stroke when treated by conventional means.
At one year, TRIOMPHE reported 90% survival free from lesion‑related death, 90% freedom from disabling stroke and 98% freedom from reintervention for endoleaks in this high‑risk cohort.
NEXUS family of stent graft systems
The NEXUS family of aortic arch stent graft systems includes NEXUS TRE™ and NEXUS DUO™.
NEXUS TRE is a triple‑branch configuration intended for use in more complex arch anatomies, while NEXUS DUO offers a dual‑branch design. The dual-branch NEXUS DUO incorporates an additional customisable, pre‑cannulated inner branch that can be configured for either the left subclavian or left carotid artery.