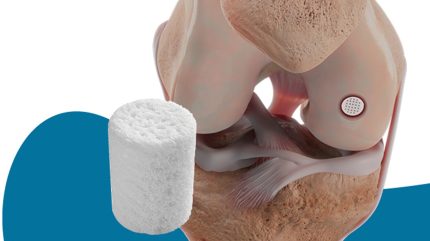
Smith+Nephew has reported that its Cartiheal Agili-C cartilage repair implant produced a higher Knee injury and Osteoarthritis Outcome Score (KOOS) compared to standard surgical care at five years in a multicentre, randomised controlled trial.
The recent findings, published in the American Journal of Sports Medicine (AJSM), indicate that patients who received the implant experienced statistically greater reductions in knee pain at two, four, and five years when compared to those receiving standard surgical care.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The study also found that these patients reported improved ability to perform daily, sport, and recreational activities across the same time points.
Researchers found consistent outcomes for patients with and without osteoarthritis, with both groups demonstrating comparable clinical results following the Cartiheal Agili-C procedure.
Approved by the US Food and Drug Administration (FDA), the Cartiheal implant is available commercially in Puerto Rico and the US.
It is composed of aragonite, a form of calcium carbonate, and serves as a biphasic scaffold to repair cartilage and restore subchondral bone.
Additionally, a Category I Current Procedural Terminology (CPT) code for the Cartiheal implant will become effective from 1 January 2027, allowing for broader procedural use.
Smith+Nephew reports that this implant’s wide indication could offer a treatment alternative to patients who previously may not have been considered for such interventions.
Last month, Smith+Nephew introduced its Allevyn Complete Care foam dressing, designed to provide advanced wound management solutions and improve pressure injury prevention.
The product features advanced technologies and a five-layer construction. Its performance is backed by clinical evidence and scientific data.
Earlier this year, the company agreed to acquire Integrity Orthopaedics, a US-based developer of the Tendon Seam rotator cuff repair system, for up to $450m.



