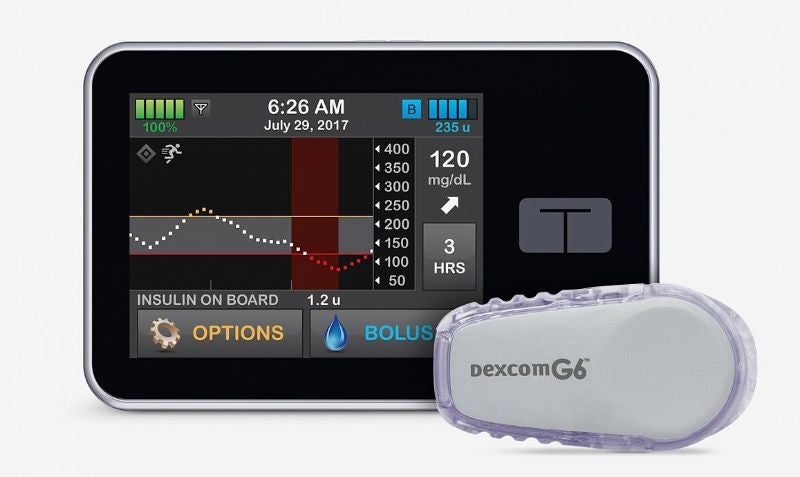
A clinical trial principally funded by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) has found an artificial pancreas system could effectively control blood glucose levels in patients with type 1 diabetes.
The closed-loop system, Control-IQ, combines Tandem Diabetes Care’s t:slim X2 insulin pump with Dexcom’s G6 continuous glucose monitor. It is designed to automatically monitor blood glucose levels and adjust insulin delivery.
The insulin pump within the system contains advanced control algorithms, which leverage a mathematical model, capturing the user’s glucose monitoring data to automatically adjust the insulin dose.
The artificial pancreas system avoids the need for fingerstick testing and daily injections or a pump.
Classified International Diabetes Closed-Loop (iDCL), the multi-centre, randomised, six-month trial assessed five different artificial pancreas clinical protocols at ten research centres in the US and Europe.
The study was the third phase in a series of trials and was performed to gain insights into the working of the system in typical daily routines of 168 participants aged 14 years and above.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataDuring the study, Control-IQ was compared to a sensor-augmented pump (SAP) therapy using a CGM and insulin pump not designed to automatically adjust insulin.
Results showed that the artificial pancreas system was more effective at controlling blood glucose levels associated with type 1 diabetes.
Participants who used artificial pancreas experienced an average of 2.6 hours per day increase in the amount of time with their blood glucose levels in the 70-180mg / dL target range, while the time did not change in the SAP group over six months.
The artificial pancreas led to improvements in time spent with high and low blood glucose, haemoglobin A1c and other diabetes control-related measurements versus the SAP group.
NIDDK director Griffin Rodgers said: “Artificial pancreas technology has tremendous potential to improve the day-to-day lives of people with type 1 diabetes.
“By making management of type 1 diabetes easier and more precise, this technology could reduce the daily burden of this disease, while also potentially reducing diabetes complications including eye, nerve, and kidney diseases.”
Tandem Diabetes has submitted the trial data to the US Food and Drug Administration (FDA) seeking marketing approval for the Control-IQ system.