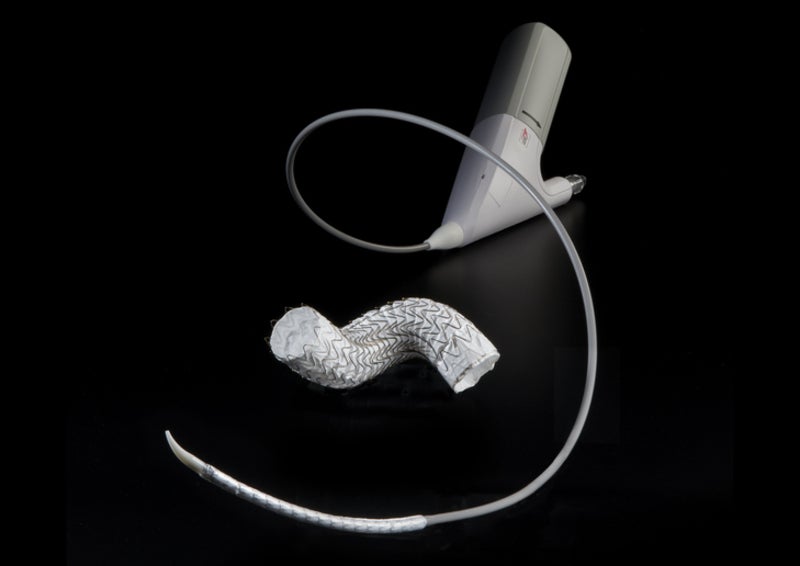
Material sciences company W. L. Gore & Associates has received approval from the US Food and Drug Administration (FDA) for its Tag Conformable Thoracic Stent Graft, a thoracic endovascular aortic repair (TEVAR) device.
The device features the Active Control delivery system to enable precise and predictable outcomes in endovascular repair of aneurysms, transections and Type B dissections of the descending thoracic aorta.
It comes with a smaller-diameter primary delivery sleeve that facilitates access to a lower profile across ten device sizes.
Gore Active Control system is said to offer controlled and two-stage deployment, with primary deployment to an intermediate diameter and secondary deployment to full diameter.
The design enables continuous blood flow throughout the deployment and also allows visualisation and refining of graft placement. It additionally allows angulation of the proximal end of the device for enhanced seal and apposition.
In addition, large device oversizing windows were designed and tested to accommodate differences in proximal and distal landing zone diameters.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataA 6% to 33% oversizing window has been engineered to help physicians choose the optimal radial force to fit patient anatomy and etiology. This could aid in the treatment of a young trauma patient or a fragile dissected aorta.
W. L. Gore & Associates vascular business leader Eric Zacharias said: “Physicians can now deploy our thoracic stent graft in the descending thoracic aorta with more operative ease, even in those patients with challenging angulated aortic arches, and meet the clinical and practical challenges of TEVAR with confidence.”
Gore Tag Conformable Thoracic Stent Graft expands the company’s endovascular products range intended for effective treatment of aortic disease.
The range also includes the Gore Excluder AAA Endoprosthesis for abdominal aortic aneurysms (AAA) and the Gore Excluder Iliac Branch Endoprosthesis to treat common iliac artery aneurysms or aortoiliac aneurysms.