Visit our Covid-19 microsite for the latest coronavirus news, analysis and updates
Follow the latest updates of the outbreak on our timeline.
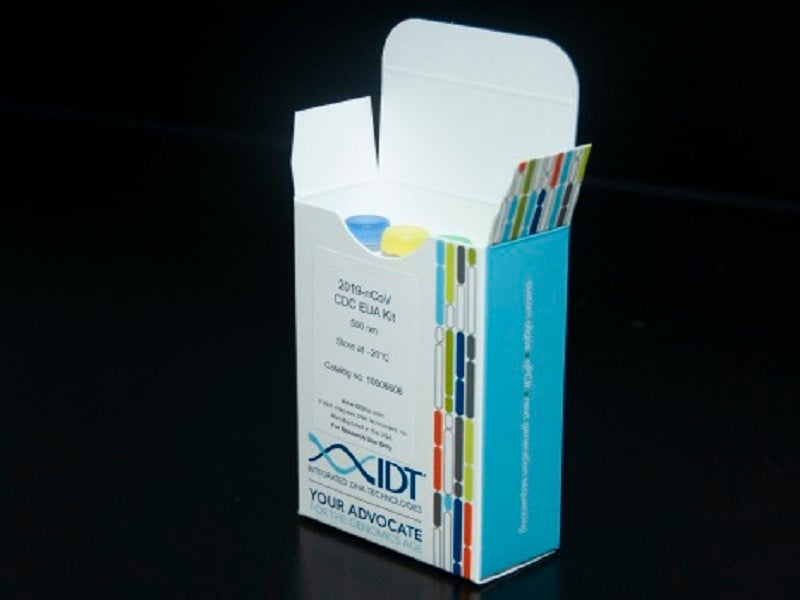
Integrated DNA Technologies (IDT) has announced its success in large-scale manufacturing of a fundamental Covid-19 component, which assists in DNA analysis of patient samples.
The company’s component is a primer and probe kit qualified by the Centers for Disease Control and Prevention (CDC).
Pursuant to the CDC Emergency Use Authorization (EUA) testing protocol, the company has shipped primer and probe kits, sufficient to enable more than one million coronavirus tests to be conducted.
Last week, IDT manufactured sufficient primer and probe kits to enable an additional 2.5 million tests.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataIt expects to manufacture five million tests per week starting from this week.
IDT President Trey Martin said: “We are honoured to be the first company in the nation to have our primer and probe kits approved by the CDC for use as a key component of the CDC EUA testing protocol for the diagnosis and detection of Covid-19.”
Previously, IDT provided products to diagnostic test manufacturers developing the test for H1N1, Ebola virus, and Zika virus.