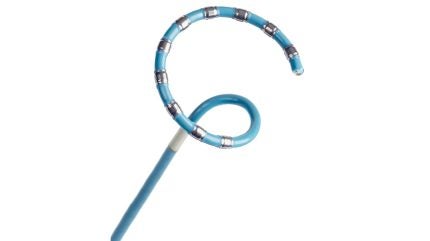
Johnson & Johnson (J&J) has launched Varipulse Pro in Europe after securing CE Mark approval, strengthening its pulsed field ablation (PFA) portfolio.
The system introduces a new pulse sequence with a lower temperature profile and an ablation that is five times faster compared to the previous sequence.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It integrates with the Carto 3 System, using advanced mapping capabilities and tissue proximity indicators to ensure precise lesion delivery.
J&J initiated initial cases under the VARIPURE multi-centre, prospective, postmarket follow-up study, aiming to generate robust evidence through this trial and other clinical studies during the commercial rollout phase.
J&J MedTech electrophysiology and neurovascular group chair Michael Bodner said: “The introduction of Varipulse Pro in Europe reflects our commitment to advancing our PFA platforms through continuous innovation, enhancing procedural experience while maintaining the consistency and precision physicians expect from the Varipulse platform.
“This launch demonstrates our dedication to continuously evolving PFA technologies based on real-world learnings and our scientific expertise, supporting physicians to deliver high-quality care and improved patient outcomes.”
The Varipulse Pro platform will be exhibited at the European Heart Rhythm Association (EHRA) annual meeting, with live case demonstrations, hands-on training sessions, and professional education.
Dr Daniel Scherr, who served as a study investigator and a consultant for J&J, will present the interim 12-month outcomes from the ongoing VARIPURE trial at the EHRA PFA Summit. Varipulse Pro is currently not approved in the US.
Last month, J&J received approval from the US Food and Drug Administration (FDA) for its TECNIS PureSee extended depth of focus (EDOF) intraocular lens (IOL), designed for use in cataract surgery.
The TECNIS PureSee IOL is expected to become available for US patients later this year.



