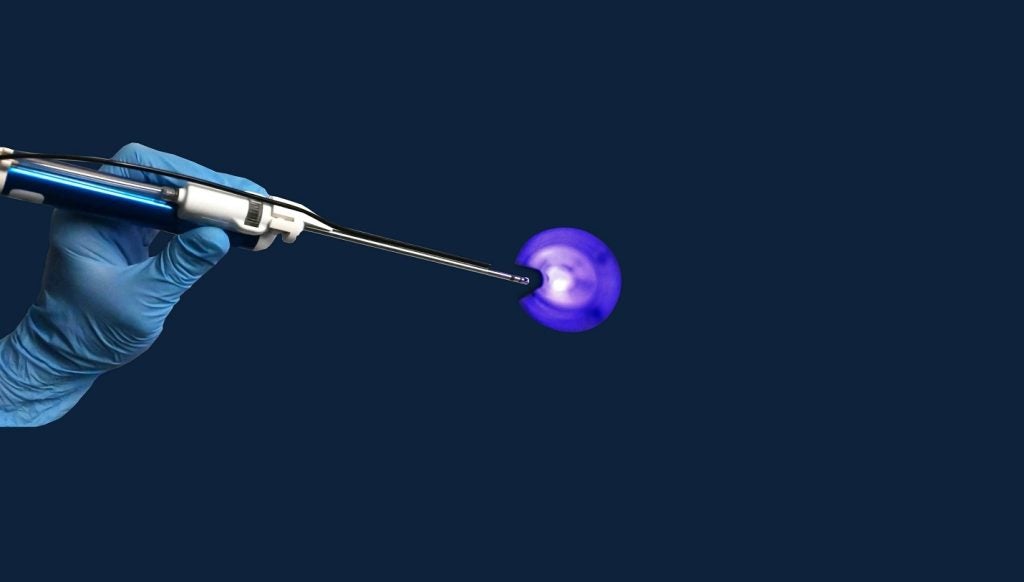
Canadian medical device firm Neovasc has announced plans to seek humanitarian device exemption (HDE) approval from the US Food and Drug Administration (FDA) for its Reducer device in certain refractory angina patients.
The European CE-marked device modifies blood flow in the heart’s circulatory system to offer relief from angina symptoms. It secured breakthrough device designation in the US in October last year.
The latest decision to seek HDE approval follows the company’s discussion with the FDA regarding the most expedient approach to potentially obtain regulatory approval in the country.
In its guidance, the US regulator suggested the HDE pathway for class IV refractory angina and/or different clinical trial designs for a broader patient population.
Neovasc intends to collaborate with the agency for the Reducer to be potentially categorised as a Humanitarian Use Device (HUD) under the HDE approval pathway for patients with the worst angina symptoms.
The company will also consider an alternate investigational device exemption (IDE) trial design for class III and IV patients.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataNeovasc president and CEO Fred Colen said: “We are encouraged by the outcome of our discussions with the FDA on the clinical evidence and the potential pathway to the US market for the Reducer.
“The FDA’s proposed alternative approaches, including a potential HDE pathway, would provide a meaningful treatment option for those patients suffering from the worst angina symptoms and who are desperate for a novel treatment in the fastest possible manner.”
Refractory angina is caused due to inadequate blood supply from the coronary arteries to the heart muscle, even when the patient is receiving standard revascularisation or cardiac drug treatments.
The condition is estimated to impact 600,000 to 1.8 million people in the US and accounts for 50,000 to 100,000 new cases annually.