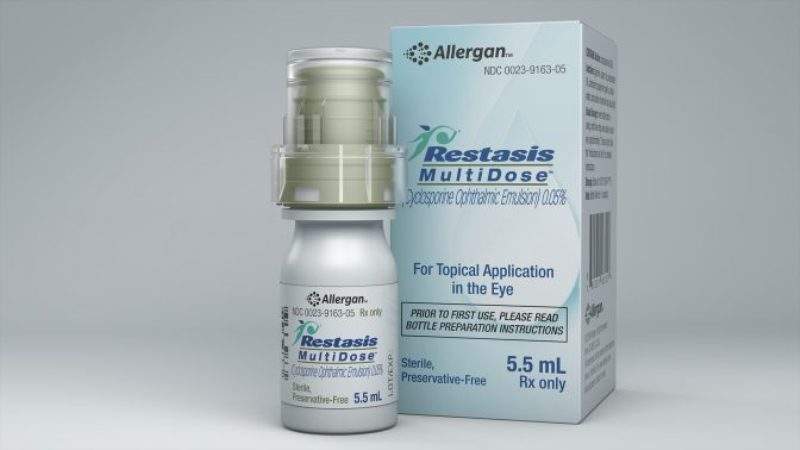

Allergan has selected Aptar Pharma’s preservative-free multidose Ophthalmic Squeeze Dispenser system for its newdry eye product, Restasis Multidose.
Restasis and Restasis Multidose are US Food and Drug Administration (FDA)-approved prescription treatmentsto help patients suffering from chronic dry eye to make more of their own tears.
The treatment is not artificial tear but is designed to help the eyes’ natural ability to produce tears.
Currently available across the US prescription market, Aptar Pharma’s patented dispenser offers improved safety to the patients.
The system, which has been approved by the FDA, can dispense accurate amount of prescribed eye treatment formulations and can maintain product integrity.
See Also:
The preservative-free nature of the system helps the dry eye patients, who use eye drops on a regular basis, which could lead to unpleasant or serious side effects.
How well do you really know your competitors?
Access the most comprehensive Company Profiles on the market, powered by GlobalData. Save hours of research. Gain competitive edge.

Thank you!
Your download email will arrive shortly
Not ready to buy yet? Download a free sample
We are confident about the unique quality of our Company Profiles. However, we want you to make the most beneficial decision for your business, so we offer a free sample that you can download by submitting the below form
By GlobalDataThe new dispenser system can removepreservativesfrom eye care medications.
Aptar Pharma president Salim Haffar said: “Our Ophthalmic Squeeze Dispenser technology has been a great success for our customers.
“The proven and unrivalled microbiological safety, combined with a precise and reproducible drop ejection allows pharmaceutical customers worldwide to enter into discussions with regulatory agencies such as the FDA.
“The ergonomic and pocket-size design and the intuitiveness of a squeezable container with a low-actuation force certainly contribute to the high levels of acceptance among patients and consumers around the world.”
Since 2012, patients and consumers in Europe, Latin America and Asia have benefited from this technology. Currently, the technology has more than 100 commercial references available on the market.
Image: Allergan’s new Restasis Multidose product in Aptar Pharma’s dispenser system. Photo: courtesy of Allergan.