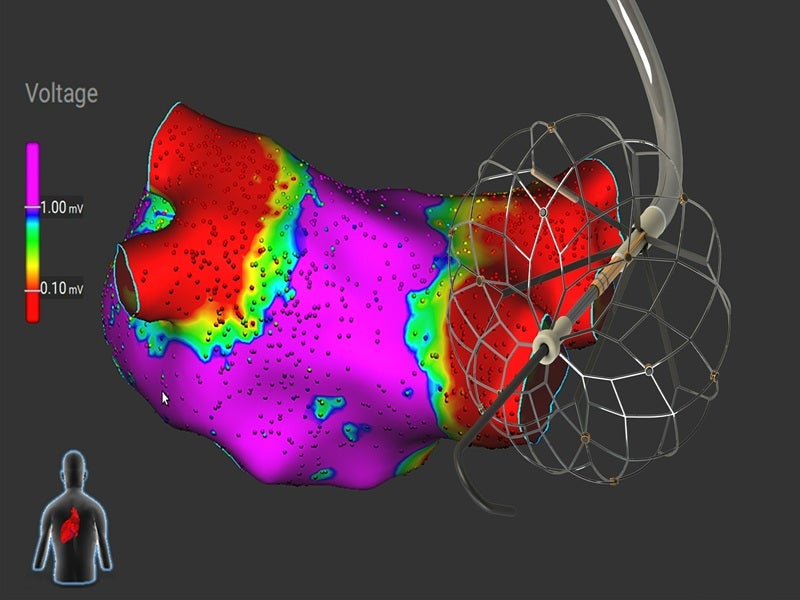
The Affera™ Sphere-360™ catheter, developed by Medtronic’s Affera, is an all-in-one mapping and single-shot pulsed field ablation (PFA) device intended for the treatment of paroxysmal atrial fibrillation (AFib).
AFib is among the most prevalent and insufficiently managed cardiac arrhythmias, impacting more than 60 million individuals globally. As the condition advances, the likelihood of severe complications, such as heart failure and stroke, as well as increased mortality risk, also rises.
The Sphere-360™ catheter was created in response to feedback from clinicians, who indicated a need for a streamlined workflow and reliable outcomes using a single catheter adaptable to variations in pulmonary vein anatomy.
The device was awarded CE Mark certification in Europe for the treatment of AFib in January 2026.
Affera Sphere-360 PFA catheter design and features
The Sphere-360 single-shot pulsed field ablation catheter is equipped with a 34mm lattice tip, which is engineered to adapt to the contours of the pulmonary veins.
This configuration enables the creation of uniform and lasting lesions in patients with atrial fibrillation, without the need to rotate the catheter, thereby supporting a more efficient procedural workflow.
The device functions as an integrated solution for navigation, mapping and ablation, requiring only a single transseptal puncture and eliminating the need for catheter exchanges.
It provides real-time local impedance data to assist in evaluating catheter-tissue proximity and facilitates access to the pulmonary veins, offering enhanced stability via its over-the-wire construction.
Designed for compatibility with the Affera mapping and ablation platform, the Sphere-360 catheter utilises an 8.5 Fr sheath, noted as the smallest available within single-shot PFA technologies.
Additionally, it can be used with the FlexCath Contour™ 10 French deflectable sheath, which is intended to simplify catheter positioning.
Affera mapping and ablation system details
The Affera mapping and ablation system is a comprehensive platform designed to enable clinicians to perform precise mapping and safe ablation, incorporating high-definition mapping and dual-energy capabilities within a single solution.
The system has been developed to meet the primary requirements of contemporary electrophysiologists, offering a range of advanced software features intended to support and streamline clinical workflows.
It is equipped with various components such as the HexaFlow™ irrigation pump, HexaGen™ RF generator, HexaPulse™ PF generator and HexaMap™ catheter interface unit.
The mapping platform supports pacing and provides real-time visualisation of compatible catheters, along with the capability to display cardiac maps in various formats.
Clinical trials on Affera Sphere-360 PFA catheter
The Sphere-360 catheter’s approval in Europe was supported by the safety and efficacy findings from the Sphere-360 study, a prospective, single-arm, multi-centre clinical trial conducted at several European locations.
In this study, more than 100 patients with AFib underwent PFA using one of three protocols: PULSE1 (n=30), PULSE2 (n=20), or PULSE3 (n=50).
Reported procedure metrics included mean procedure time of 57.9 ± 20.6 minutes, left atrial dwell time of 22.2 ± 11.8 minutes and fluoroscopy time of 6.8 ± 5.7 minutes.
All 395 targeted pulmonary veins were successfully isolated, with a mean pulmonary vein isolation (PVI) time of 11.5 ± 6.0 minutes and an average of 4.0 ± 1.3 lesions per vein.
At the one-year follow-up, 82% patients in the overall cohort and 88% of patients in the PULSE3 group remained free from arrhythmia recurrence.
The PVI durability cohort was 98% per vein and 93% per patient, with no safety events reported among those treated with the optimised waveform protocol.
PVI durability with PULSE3 (n=40) was 98% (per-vein) and 93% (per-patient).
No primary safety events, defined as serious device-related incidents within seven days post-ablation, were observed in the trial.
Additional trials
The Horizon 360 IDE study is a prospective, single-arm, unblinded pre-market clinical investigation that commenced in January 2026 at multiple sites across the US.
The study is designed to assess the safety and effectiveness of the Sphere-360 catheter in conjunction with the Affera mapping and ablation system for the treatment of AFib.
Up to 300 participants are expected to be enrolled at as many as 26 locations in the US to evaluate the study’s primary objectives of freedom from atrial tachyarrhythmia recurrence 12 months post-ablation.