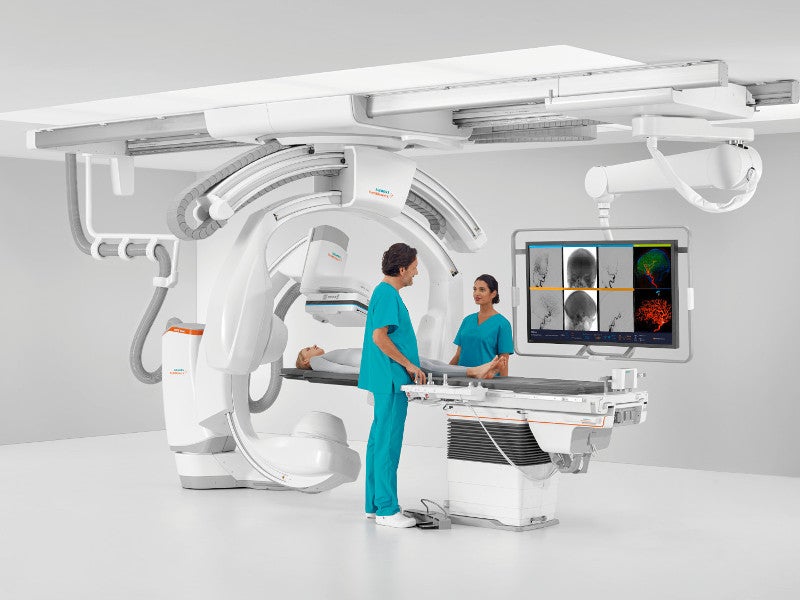
ARTIS icono is a biplane angiography system developed by Siemens Healthineers for single or biplane diagnostic imaging and interventional treatment of patients including obese and paediatric patients.
The system performs various procedures such as neuro-angiography, general angiography, cardiac angiography, rotational angiography, multipurpose angiography, cardiology, body vascular interventional radiology (IR) as well as whole-body radiographic procedures.
ARTIS icono performs image-guided surgery involving X-ray, image fusion and navigation systems, for use in the operating room.
The US Food and Drug Administration (FDA) received an application for 510(k) clearance of the system in August 2019. The system received FDA clearance in September 2019. It was first installed in the UW Health’s University Hospital in Madison, Wisconsin, in February 2020.
In July 2022, ARTIS icono ceiling angiography system received FDA clearance for its use in a wide range of routine and advanced procedures in interventional radiology and cardiology.
Siemens Healthineers launched the ARTIS icono angiography system in India in September 2022.
ARTIS icono angiography system design and features
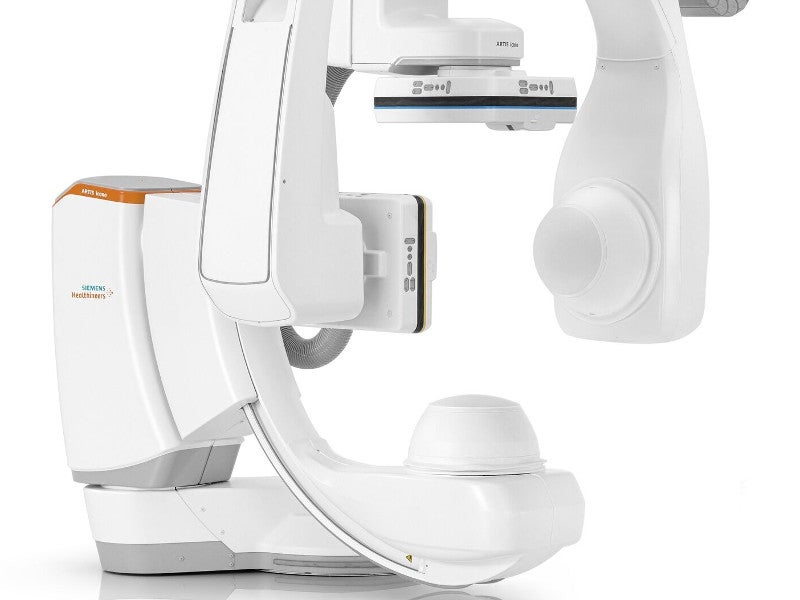
ARTIS icono is a modular, high-precision angiography system, for two-dimensional and three-dimensional imaging. It facilitates the visualisation of the blood vessels in the body.
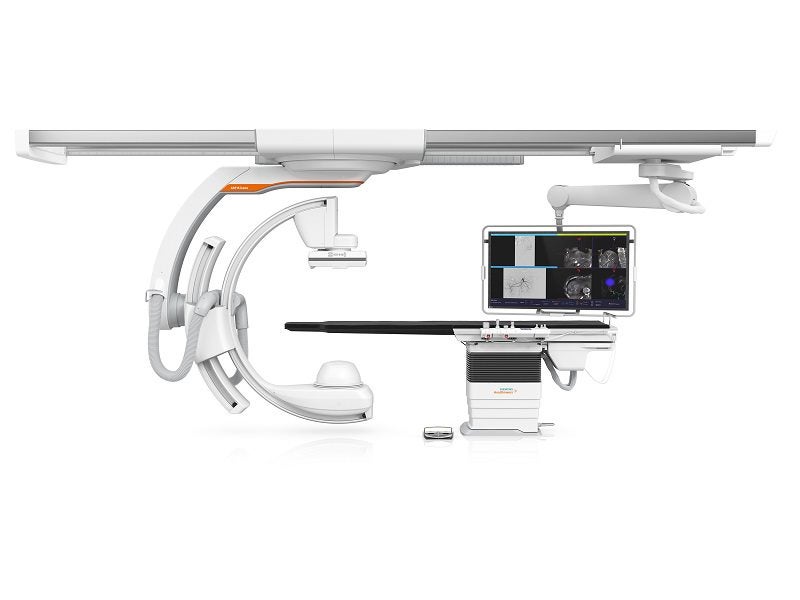
ARTIS icono configuration works in two different ways, biplane or floor for delivering specialised angiography systems.
The system comprises a patient table, a full motorised C-arm with multiple axes, display suspension system, footswitch for releasing radiation and control console.
The display attaches on a movable display suspension system at the ceiling, revealing the X-ray images, while the X-ray generator is at a separate location. Physicians operate the system by the tableside control module, enabling them to move the table and C-arm accordingly for reliable imaging. Catheters and additional devices are also adjustable during X-rays.
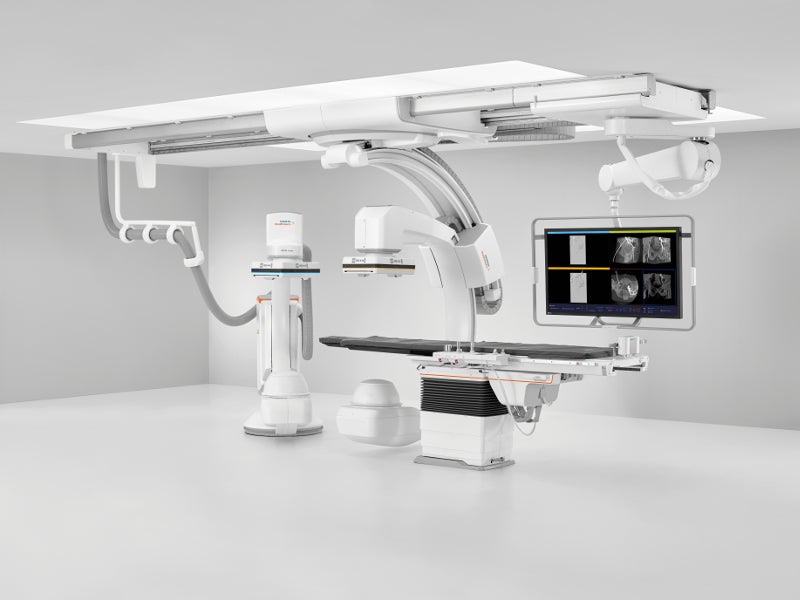
ARTIS icono floor has a small footprint, allowing easy installation. It assists in various vascular IR procedures with patient coverage of 2.10m and unparalleled lateral coverage of 1.90m. The C-arm can mount on the floor or the ceiling for the biplane systems.
The system also comprises a flat-panel detector, X-ray tube, collimator, and image post-processing. The control room equipped with monitors and a footswitch location enables post-processing.
In February 2022, Siemens Healthineers introduced the ARTIS icono biplane with new detectors designed for detecting and diagnosing cardiovascular conditions such as cardiac arrhythmia and coronary heart disease.
The system simplifies clinical workflows and offers excellent image quality at a low radiation dose. It enables simple positioning of the C-arm to display images at steel angulations for complex cardiovascular diseases.
ARTIS icono ceiling angiography system
ARTIS icono ceiling angiography system enables early diagnosis and treatment decision for precise tumour embolisation. The new ceiling-mounted system helps in performing super-selective procedures and other complex interventions.
ARTIS icono ceiling enables 3D imaging at the patient’s head as well as side over a complete angular range of 200 degrees. The system features new controls and drives for the C-arm to enable fast rotation and exact system movements. The 3D images can be acquired in less than three seconds, which helps in reducing motion artefacts.
ARTIS icono angiography system software
ARTIS icono has been modified with software VE2, enhanced with algorithms by architecture and organ programme parameters for improved image quality and workflow acceleration. The software also updated the automatic exposure control and ClearStent Live, a real-time stent positioning verification tool.
The system can be integrated with Syngo application software such as Syngo Dyna3D HighSpeed, Syngo Dyna3D Sine Spin, TwinSpin and Syngo Dyna3D Multiphase for enhanced 3D imaging in both configurations.
2D imaging has been improved by the new OPTIQ imaging chain, which optimises the image quality by balancing the radiation dose to the patients and physicians.
Case Flows tool standardises the execution of operation procedure across various ARTIS icono laboratories.
Details of software features
In 3D imaging, Syngo Dyna3D HighSpeed reduces the required time for low-contrast 3D imaging from 20 seconds to eight seconds, resulting in CT-like images with nominal disturbance from moving artefacts.
TwinSpin facilitates easy switching between 2D biplane imaging and 3D imaging, accelerating workflow. It lowers the risk of collision of the equipment.
Syngo Dyna3D Sine Spin allows better determination of bleeding regions by reducing the artefacts around the skull cap and cranial base. Syngo Dyna3D Multiphase allows identification of the areas in the brain with lowered blood flow by producing time-resolved CT volumes without patient movement to the CT scanner.
The Lateral Plane switch enables change in detector position from the right side to the left side of the patients within 90 seconds.
Key benefits of ARTIS icono
The system broadens precision medicine with enhanced structure visualisation, including the difficult-to-delineate structures such as cranial base and skull cap. It assists in stroke diagnosis, improving the therapeutic options for the patients.
The ARTIS icono biplane allows rapid axial movements with high flexibility for multidisciplinary laboratory usage.