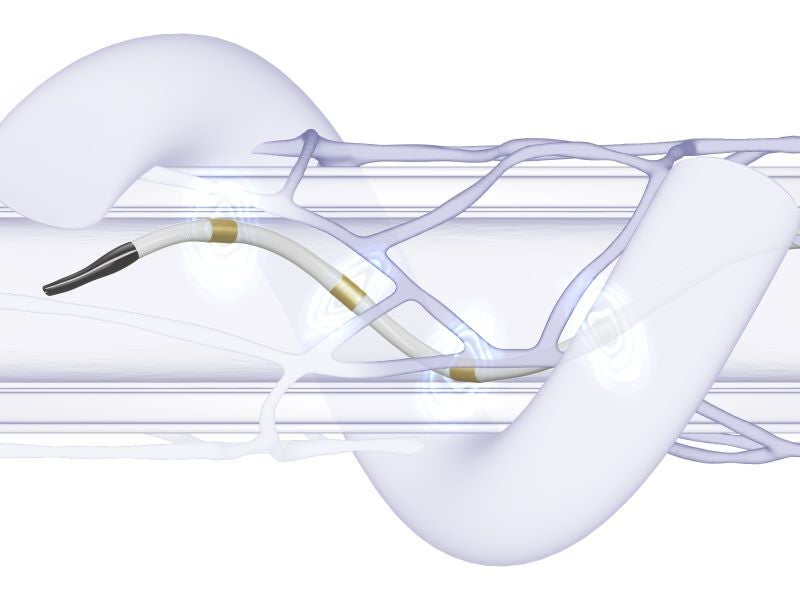
The Symplicity Spyral™ renal denervation (RDN) system, also known as the Symplicity blood pressure procedure, is a minimally invasive method for the treatment of hypertension.
Developed by US-based medical device company Medtronic, the system is currently approved in 77 countries worldwide.
The device provides patients with a new supplementary method for reducing blood pressure. It is clinically proven to help reduce blood pressure by using radiofrequency (RF) energy in nerves near the kidneys. The nerves can become overactive and play a role in the development of high blood pressure.
Hypertension, commonly referred to as high blood pressure, is the underlying cause of stroke, heart attacks and death. In 2024, an estimated 1.4 billion adults worldwide aged between 30 and 79 were living with hypertension, equivalent to around 33% of people in this age group.
Approximately 80% of adults do not have hypertension under control. Lifestyle modifications and/or medication have historically been the sole viable methods for addressing high blood pressure.
Regulatory approvals
The Symplicity Spyral RDN system was granted the US Food and Drug Administration’s (FDA) breakthrough device designation in 2020. The FDA approved the RDN system in November 2023.
The system received approval from Health Canada in March 2024, followed by the National Medical Products Administration in China in May 2024.
Japan’s Pharmaceuticals and Medical Devices Agency approved the device in September 2025.
Symplicity Spyral RDN system procedure details
The Symplicity™ blood pressure procedure involves inserting a single-use Symplicity Spyral multi-electrode renal denervation catheter into the artery that leads to the kidney, after sedating the patient. The doctor then administers precisely controlled and targeted RF energy to the renal nerves, using the Symplicity G3 renal denervation RF generator.
The RF energy effectively interrupts the excessive sympathetic signalling that occurs between the kidneys and the brain, leading to a drop in blood pressure while ensuring safety.
After the procedure, the implant is safely removed to leave no traces behind.
The procedure is safe and effective, providing significant and sustained blood pressure reductions. It is subsequently an optimistic treatment for clinicians and patients who are in desperate need of reducing their blood pressure.
Symplicity Spyral RDN catheter details
Medtronic’s Symplicity Spyral RDN catheter features an easy-to-use, rapid-exchange, plug-and-play design.
The distal self-expanding array of four electrodes on the catheter is guided to the treatment site using a 0.014in guidewire. The catheter can effectively navigate intricate anatomy and treat a wide range of complex anatomy vessels ranging between 3mm and 8mm in diameter.
It employs a helical electrode arrangement to simultaneously ablate all four quadrants of the vessel, allowing for the denervation of the main artery, accessory vessels, and branch vessels to enhance the likelihood of achieving complete denervation.
A non-occlusive catheter design permits natural blood flow for arterial wall irrigation and cooling. Additionally, it is suitable for use with a 6Fr guiding catheter.
Symplicity G3 generator features
Utilising a real-time responsive algorithm, the Symplicity G3 generator automatically regulates power delivery to each electrode based on temperature and impedance sensor feedback to ensure safe energy distribution. Individual electrodes can be selected or deselected based on the ability to make full contact with the vessel wall.
The RF power output per electrode can reach a maximum of 6.5W (26W total), delivering energy when the measured impedance falls within the range of 175 ohms to 1,200 ohms.
Clinical trials on Symplicity Spyral RDN system
The FDA’s approval of the RDN system follows ten years of clinical research and development of the Medtronic RDN technology.
The SPYRAL HTN Global Clinical Programme, the most comprehensive clinical programme studying RDN, involves more than 25,000 patients from 25 clinical centres in Australia, Austria, Germany, Greece, Japan, the UK and the US. The trials were conducted with and without medication and in patients with high baseline cardiovascular risk.
The long-term efficacy and safety of the device in the presence of antihypertensive drugs were studied in the SPYRAL HTN-ON MED clinical trial, which was a randomised, single-blind, sham-controlled trial.
The SPYRAL HTN-ON MED clinical trial showed clinically significant, safe and sustained blood pressure reductions through three years.
RDN patients experienced a decrease of 18.7mmHg in 24-hour systolic ambulatory blood pressure (ABPM) compared to the baseline, and a 20.9mmHg reduction in office systolic blood pressure. More than 80% of patients on medication achieved a systolic blood pressure (SBP) below 140mmHg with the Symplicity procedure.
Results from the Medtronic-led patient preference study indicate that approximately one-third of patients were likely to choose the interventional treatment due to its proven blood pressure reduction results.
The SPYRAL HTN-OFF MED pivotal trial without medications demonstrated noteworthy reductions in blood pressure levels after three months. Specifically, a decrease of 9.2mmHg in office SBP and a 4.7mmHg decrease in 24-hour ABPM were noted.
The trial demonstrated that the Symplicity blood pressure procedure achieved greater reductions in blood pressure compared to the control group after three months. Its benefits in decreasing blood pressure are continuous, and the system provides around-the-clock protection, including during the high-risk early hours of the day.
The long-term data from the trial at three years, announced in October 2025, showed significant and sustained blood pressure reductions in both office-based and 24-hour ABPM.
RDN patients showed a 14mmHg reduction in 24-hour ABPM compared to 9.3mmHg in the sham group and an 18.5mmHg reduction in office-based SBP compared to 11.7mmHg in the sham group.
Additional clinical trials
The SPYRAL HTN clinical programme also includes the SPYRAL AFFIRM prospective, international, multicentre, single-arm trial, which is designed to assess the safety and effectiveness of the Symplicity Spyral RDN system in a real-world population of 987 patients across 100 sites, with more than half of participants based in the US.
The initial results of the trial after six months were announced in October 2025, along with the completion of the enrolment for the study.
Data from a cohort of 210 patients from 59 sites across Europe, Australia and the US demonstrated significant reductions in both home and office SBP across multiple high-risk subgroups, including those with isolated systolic hypertension (ISH), diabetes mellitus (DM) and chronic kidney disease (CKD), with no increase in anti-hypertensive medication use.
Patients in the DM group experienced a reduction of 13.3mmHg in office-based SBP and 5.9mmHg in home-based SBP, while patients in the ISH group experienced a reduction of 10.6mmHg in office-based SBP and 4.6mmHg in home-based SBP.
CKD group patients experienced a reduction of 19.3mmHg in office-based SBP and 5.4mmHg in home-based SBP.
Additionally, the number of prescribed anti-hypertensive medications remained stable across all groups, and no major safety events were observed, further validating the safety of the Symplicity blood pressure procedure in high-risk patients.
Global SYMPLICITY Registry (GSR-DEFINE) is another trial designed as a prospective, all-comers observational study, spanning 251 sites in 55 countries and targeting 5,000 patients worldwide.
The trial builds on the Global SYMPLICITY Registry, the largest real-world dataset assessing the long-term safety and performance of the Symplicity Spyral RDN system in patients with uncontrolled hypertension.
The registry demonstrated a 16.3mmHg reduction in office-based SBP at three years among 1,450 patients enrolled outside the US.