No Filter Selected
No Filter Selected
No Filter Selected
No Filter Selected

Fast, Expert, Open: Lawrence Tallon’s vision for the MHRA
Lawrence Tallon on priorities as MHRA CEO, the evolution of UK medical device regulation and the agency’s future leadership.

Podcast: MHRA CEO Lawrence Tallon on the future of medical device regulation in the UK
Lawrence Tallon, CEO of the MHRA, discusses the agency's initiatives to advance the UK's regulatory protocols for medical devices.

MHRA secures $4.8m to expand AI medical device programme
AI Airlock is jointly run by the MHRA, DHSC, NHS AI Team, and Team AB, the consortium of UK Approved Bodies.

Neurovalens receives EU MDR compliance for sleep insomnia device
Achieving MDR compliance shows Modius Sleep meets safety, clinical, and quality standards required for entry into the EU market.

LifeVac receives FDA De Novo classification for anti-choking device
LifeVac is approved for use as a single-use, non-powered, non-invasive airway clearance device.

Daiichi and GAIA join forces to commercialise digital cardiovascular therapy
Pharma companies are increasingly combining the strengths of pharmaceutical and digital health interventions to improve outcomes.

Medical device testing in UK rises amid global competitiveness push
This increase in activity comes as the UK looks to enhance its standing in the medtech sector as global competition mounts.

MHRA launches consultation on UK’s long-term approach to CE-marked medical devices
One of the key proposals the MHRA seeks feedback on is the ‘indefinite recognition’ of EU CE-marked medical devices in the UK.

UK government champions healthtech through new NHS access programme
The National Healthtech Access Programme aims to improve accessibility to healthtech products across the NHS.

MHRA outlines upcoming IVD submission shift for clinical trials
From 28 April, IVDs used clinical trials will be subject to an upfront assessment by the MHRA as part of a Clinical Trial of an Investigational Medicinal Product submission.

New MHRA resources prompt clarity on use of digital mental health tools
The MHRA’s new resources are intended to help individuals make better-informed decisions about the digital mental health support tools they use.

The importance of real-world evidence in transforming healthcare decision-making
Real-world evidence (RWE) empowers payers and providers to make more informed, cost-effective treatment and coverage decisions by addressing key knowledge gaps.
UK reforms highlight the case for digital submissions in medical device regulation
Diogo Geraldes, director of regulatory strategy, Europe at Veeva MedTech, discusses the potential of digital submissions to benefit manufacturers, regulators, and patients.

Wellcome backs MHRA and NICE’s digital mental health tech push with £2m funding
The funding from the Wellcome Trust will aid the entities in continuing their work on developing safety protocols around digital mental health technologies.

The transatlantic medtech investment gap: why does it exist and can it be plugged?
Ivor Campbell, CEO of medtech recruitment consultancy Snedden Campbell, reflects on what companies in the UK and Europe can do to compete for investment against the US.

NHS trials rapid blood test to diagnose serious infections in children
The project investigating the blood test is set to continue until March 2026.

Medtech devices becoming “learning systems”, says Google Cloud exec
As agentic AI advances, the technology holds the potential to make medtech systems and devices more anticipatory.

MHRA establishes commission to advance AI deployment across the NHS
By rewriting the rulebook to clarify regulatory uncertainty around AI’s use in healthcare, the MHRA aims to position the UK as a global hub for health tech investment.
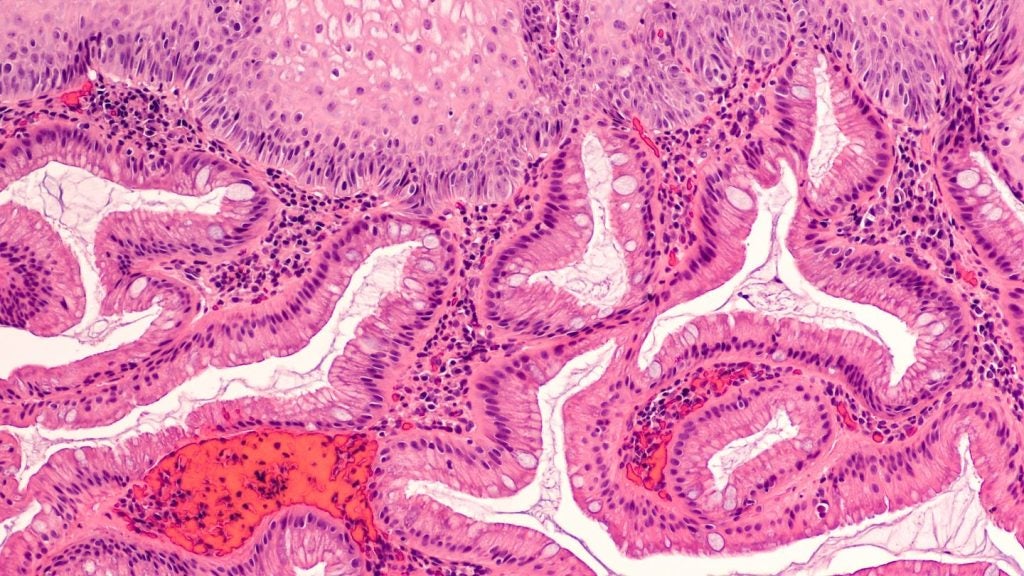
Cyted raises $44m to propel US market expansion of its endoscopic capsule
The British company plans to use the funds to drive commercialisation efforts for its GI diagnostics platform and expand its portfolio of tests.

Health institutions to have say in MHRA’s reforms for in-house manufactured medtech
The MHRA aims to refine a 2021 policy that exempts medical devices made and used by health institutions from certain requirements.
