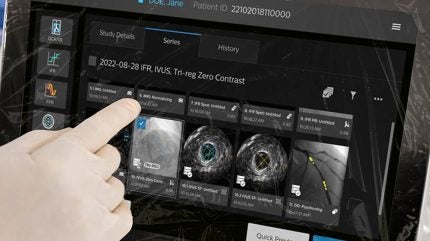
Philips has launched the IntraSight Plus coronary interventions platform, which is cleared for clinical use in Europe and the US.
With the US Food and Drug Administration (FDA) 510(k) clearance and CE marking, the interventional guidance system aims to streamline access to diagnostic and treatment planning tools for clinicians.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Philips stated that traditionally, interventional cardiologists have depended on angiography alone for percutaneous coronary interventions (PCI), a minimally invasive procedure.
Current guideline-supported tools such as intravascular ultrasound and physiology are often delivered through separate systems, which can hinder adoption in busy catheterisation laboratories.
Philips’ IntraSight Plus aims to address these challenges by providing a single integrated solution that can reduce system operation time by up to 47% and support bedside control.
During this month, Dr Jan-Malte Sinning treated the first patient with IntraSight Plus at Cellitinnen-Krankenhaus St Vinzenz in Köln, Germany. The hospital recently upgraded its catheterisation laboratory with Philips Azurion interventional suites.
The integration of IntraSight Plus into this environment aims to enhance workflow for physicians.
Using both Azurion and IntraSight Plus, Dr Sinning performed complete revascularisation before discharge.
IntraSight Plus merges Philips’ existing technologies such as IntraSight and SyncVision, offering features that include co-registration, instantaneous wave-free ratio/fractional flow reserve (iFR/FFR) physiology, Class IA IVUS, and real-time device visualisation within one system.
Philips’ image-guided therapy devices business leader Stacy Beske said: “As more advanced technology is integrated into the cath lab, the need for connectivity between the imaging tools is more important than ever before.
“IntraSight Plus was built using feedback from our customers and represents a fundamental shift in interventional cardiology – a future driven by integrated intelligence that lets physicians focus on the decision-making that only they can do.”
Before this, the FDA cleared Philips’ SmartHeart AI, providing clinicians with a tool that streamlines cardiac magnetic resonance workflows.



