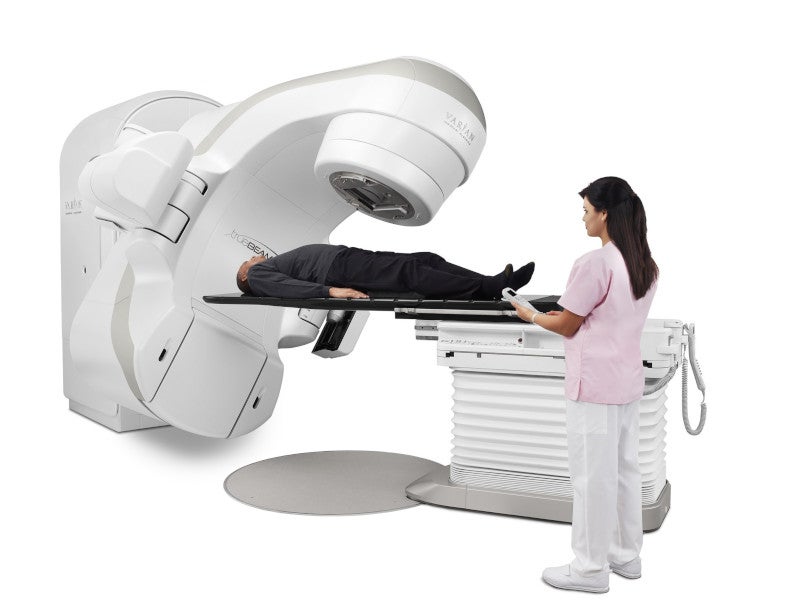
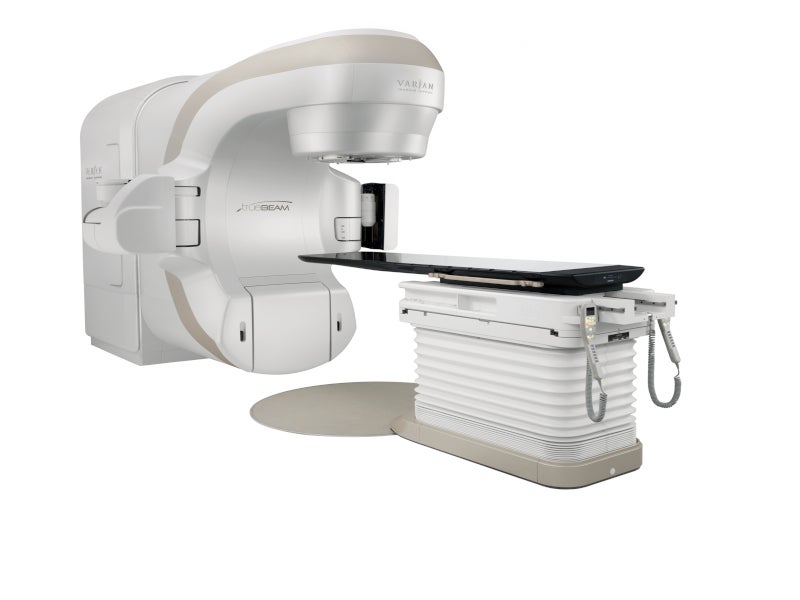
TrueBeam™, an advanced radiotherapy system by Varian Medical Systems, a US-based medical device company and part of Siemens Healthineers, was developed to deliver powerful and precise cancer treatments.
The medical linear accelerator excels in image-guided stereotactic radiosurgery and radiotherapy, targeting tumours and lesions throughout the body including the lungs, breasts, and head and neck areas.
Varian Medical Systems introduced the TrueBeam system in April 2010.
Regulatory approvals
TrueBeam gained marketing approval in China in April 2011 and in Japan in June 2011.
The US Food and Drug Administration (FDA) approved the device for clinical use in August 2011. The device also carries a Conformité Européenne mark of European conformity and is approved by Health Canada.
The system expanded its global reach with regulatory clearance in India in November 2012.
In February 2024, Varian received 510(k) clearance from the FDA for the TrueBeam system featuring the HyperSight imaging solution.
TrueBeam secured FDA 510(k) clearance in March 2026 for the delivery of low-dose radiation therapy in adults with medically refractory osteoarthritis.
TrueBeam radiotherapy system design and features
The TrueBeam radiotherapy system, comprising a beam-producing mechanism and an external control panel, is designed to generate photon, electron and diagnostic X-ray radiation. Installed within a shielded vault in medical facilities, the beam-producing unit ensures safety from radiation, while the control panel, equipped with dedicated software, is stationed outside the treatment area.
Equipped with six rotational couches, a motion management interface and a visual coaching tool, the system boasts a comprehensive cone-beam computed tomography (CBCT) imaging suite, featuring 4D CBCT, gated CBCT, short ARC CBCT, extended-length CBCT and multi-scan CBCT capabilities. Its advanced design incorporates cutting-edge imaging and motion management technologies.
Crucial for tumours affected by respiratory motion, such as those in the thoracic region, TrueBeam’s respiratory gating technology aligns radiation delivery with the patient’s breath cycle, targeting the tumour precisely when it is ideally positioned. The technology opens up new possibilities for treating cancers in the lung, breast, prostate and head and neck, among others.
TrueBeam’s ability to seamlessly integrate external solutions, hardware, innovative advancements, safety features, software and treatment protocols provides care teams with transformative technology. The integration fosters collaboration, enabling clinics to enhance treatment offerings, extend their services and accelerate the adoption of new healthcare initiatives.
TrueBeam radiotherapy system details
The system harmonises components, creating a seamless interplay that ensures unparalleled accuracy. Imaging, patient positioning, motion management, beam shaping and dose delivery come together in a well-orchestrated sequence that leaves no room for error.
Equipped with LaserGuard II, the device uses 2.5MV and 4–25MV energy for operation. It also integrates the previously FDA-cleared Trilogy radiotherapy system and related accessories.
The kilovolt (kV) radiation source features touchguards and a supplemental capacitive collision detection system. It enables proximity detection along with a kV detector and positioning units.
The system includes various intelligent tools such as HyperArc radiosurgery and RapidArc radiotherapy, allowing an advanced treatment option for cancer patients.
Intelligent tools details
HyperArc high-definition radiation therapy, an advanced intracranial stereotactic radiosurgery solution, is employed by Varian’s TrueBeam and Eclipse™ platforms. It enables treatment delivery with a single click and features pre-set imaging waypoints for complex procedures.
RapidArc, also known as Volumetric Arc Therapy, administers a 3D dose to the entire tumour with high precision in under two minutes, utilising a 360° rotating gantry in single or multiple arcs.
Gated RapidArc radiotherapy precisely targets lung and other respiratory motion-affected tumours by compensating for movement during the patient’s breathing, operating in a continuous rotation of the treatment machine.
The system incorporates iterative CBCT technology, an advancement over conventional CBCT. It leverages Acuros® computed tomography scatter to reduce scatter and enhance image quality, particularly in visualising soft tissue in the pelvis, head and neck. Integration of the system with the ARIA oncology information system and the Eclipse treatment planning system streamlines the treatment planning process for clinicians.
TrueBeam’s HyperSight imaging solution enhances accuracy in radiotherapy treatment delivery by enabling high-quality image acquisition during daily treatments, ensuring precise tumour targeting and healthy tissue conservation.
HyperSight allows linear accelerators to generate images with Hounsfield Unit accuracy for treatment planning directly from acquired CBCT images, which facilitates offline adaptive planning, accommodating anatomical changes in the tumour and surrounding organs without a separate CT scanner.
With TrueBeam, HyperSight captures images for all anatomical sites with a 50% faster gantry rotation, reducing acquisition time significantly compared to traditional CBCT scans, which can take up to 60 seconds. The swift process decreases motion-related artefacts and potentially reduces patient discomfort and anxiety.
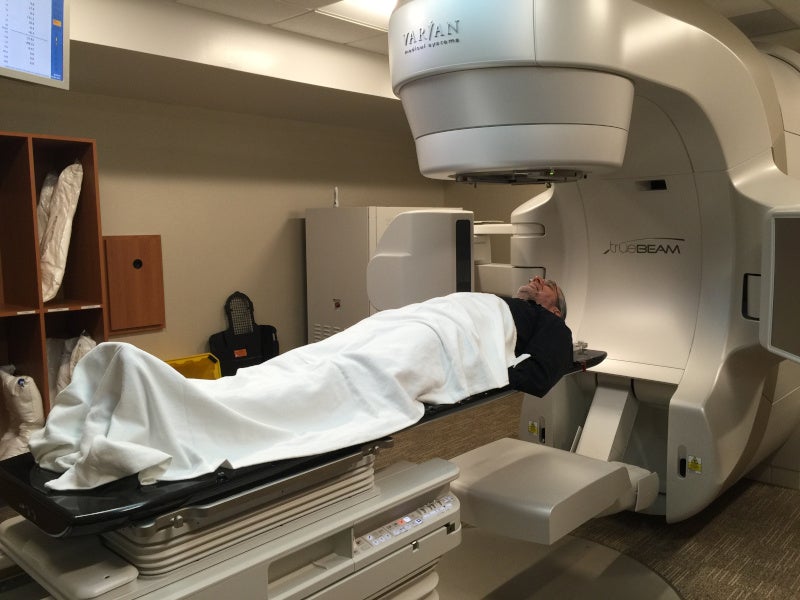
Treatment using TruBeam
Treatment using TrueBeam is a three-step process, which comprises planning, treatment preparation and radiation treatment.
It starts with capturing intricate details of the tumour and its surroundings. The images serve as a map for the specialists to navigate, and the experts devise a personalised radiation therapy plan accordingly.
It includes determining the precise doses of radiation, calculating the number of sessions required, selecting the optimal radiotherapy modality and designing an approach that minimises impact on healthy tissues.
For treatment preparation, the patients are positioned using specially moulded devices. The treatment area is carefully marked with permanent ink to guide the focus of the radiation.
The equipment is calibrated and aligned for motion monitoring. The parameters are meticulously adjusted to guarantee the utmost accuracy in radiation delivery.
In the radiation treatment phase, the radiation doses are delivered for a specific period in controlled doses. The TrueBeam STx linear accelerator rotates around the patient to deliver the radiation beams. It delivers calculated radiation beams carefully, with shaped and reshaped beams in real time, targeting the tumour from multiple angles. A cone-beam CT, a form of CT, is used to administer 25% less radiation.
TrueBeam accurately administers radiation by capturing images of the tumour, verifying its positional accuracy every ten seconds and making necessary adjustments to the radiation beams in case of tumour position changes.
Benefits of the TrueBeam radiotherapy system
The TrueBeam radiotherapy system enables patients to be treated in significantly fewer sessions, ranging from one to five, as opposed to the traditional 20–40 sessions, depending on the tumour type. The reduction in sessions diminishes the likelihood of side effects for patients.
The system offers a swift and straightforward treatment process, reducing procedure times from the previous ten to 30 minutes to approximately two minutes.
TrueBeam’s dynamic imaging capabilities, encompassing 2D, 3D and 4D imaging, allow for real-time customisation and synchronisation of planned treatments with the patient’s condition during radiotherapy.
Clinicians benefit from the motion management interface, which provides precise information on the treatment delivery location, thereby enhancing the accuracy and precision of the treatment. The system’s CBDT technologies improve the imaging of moving targets such as those in the lungs and abdomen, which is instrumental in advancing breath-hold stereotactic body radiation therapy (SBRT) and extracranial radiosurgery.
The TrueBeam system is also versatile, supporting a variety of radiation treatments including intensity-modulated radiotherapy, SBRT and RapidArc radiotherapy.