In the field of cancer imaging, advancements in artificial intelligence (AI) have brought forward a new era of precision and efficiency. AI-powered technologies are changing the way we detect and diagnose cancer, offering immense potential for improving patient outcomes.
New emerging technologies are constantly hitting the market from breast scans to gastric scans. But how are these technologies impacting the professionals working in the field and how will regulation change the future of AI?

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
At present, government bodies are planning regulatory rules for the use of AI but that doesn’t mean they are not in support of the new tools. Many initiatives and investments are being put forward by government bodies to support the use of AI.
One of the most significant challenges in the battle against cancer is early detection. Timely identification of cancerous cells greatly increases the chances of successful treatment. AI algorithms have shown remarkable capabilities in analysing medical images, such as mammograms, computed tomography (CT) scans, and Magnetic Resonance Imaging (MRIs), to detect subtle abnormalities that might be missed by human observers who suffer from workplace stress or fatigue.
These machine learning algorithms analyse images and data, highlighting areas of interest for investigation helping radiologists and oncologists identify anomalies giving them the capability to save countless lives by early detection.
Government support
In the UK the government pledged a £21 million fund to roll out artificial intelligence across the National Health Service (NHS) – giving NHS staff the latest AI technology to diagnose and treat patients more efficiently.
On average 600,000 chest X-rays are performed each month in England, and with the distribution of diagnostic AI tools, NHS Trusts clinicians will have the ability to diagnose cancer patients earlier on.
Eager about the fund and its potential to improve cancer imaging Dr Katharine Halliday, President of the Royal College of Radiologists said: “There is huge promise in AI, which could save clinicians time by maximising our efficiency, supporting our decision-making, and helping identify and prioritise the most urgent cases. Together with a highly trained and expert radiologist workforce, AI will undoubtedly play a significant part in the future of diagnostics.”
By incorporating AI into the diagnostic workflow, radiologists can obtain additional insights and validation, reducing the occurrence of false positives and false negatives. This collaboration between human expertise and AI assistance enhances diagnostic confidence and helps ensure that patients receive the most appropriate care.
There have also been other investments in AI tools. “The UK government has recently invested £16 million; nine companies were given funding through the AI in Healthcare Awards. The winners included AI systems that help detect cancer, diagnose rare diseases, identify women at high risk of premature birth, and support the treatment of neurological conditions like dementia,” said Alexandra Murdoch, medical analyst at GlobalData.
In the EU the European Commission has rolled out its Europe’s Beating Cancer Plan: Launch of the European Cancer Imaging Initiative. Creating a digital infrastructure connecting resources and databases of cancer imaging data across the EU. The project is starting with 21 clinical sites from 12 countries and includes two projects, the EUCAIM project and the AI Testing and Experimentation Facility for Health.
In North America, the US and Canadian governments are investing in AI initiatives too. “I think any initiative that aims to help healthcare professionals ease their workload while also improving cancer screening is a good one.” Sharing her thoughts on Europe’s Beating Cancer Plan Murdoch added, “I do think that they may run into a few issues along the way because there are not yet a lot of initiatives like this, so they are kind of paving the way so to speak. But I think we will see a lot more of these types of programs in the future,”.
Products in the pipeline
Many products are saving time by reducing processes. For example, US based full-body MRI screening company Ezra who secured 510(k) clearance from the US Food and Drug Administration (FDA) for its new AI technology, Ezra Flash can now facilitate a 30-minute full body MRI scan while decreasing the cost by 30% according to Ezra CEO Emil Gal.
Provider of breast CT technology, Koning Corporation, has integrated AI into its imaging platform to produce high-resolution 3D images they claim can assist early detection and treatment of breast cancer.
There are also big-name collaborations such as Microsoft who partnered with digital pathology company Paige to launch a suite of AI tools, Paige Breast Suite, to aid pathologists in breast cancer diagnosis.
These are just a few of the innovations happening in the cancer imaging space and at the rate of AI development new products are constantly emerging in the market. According to a GlobalData report, AI is growing in several fields globally, and the market anticipated to reach $93 billion in 2023, up 12% from 2022.
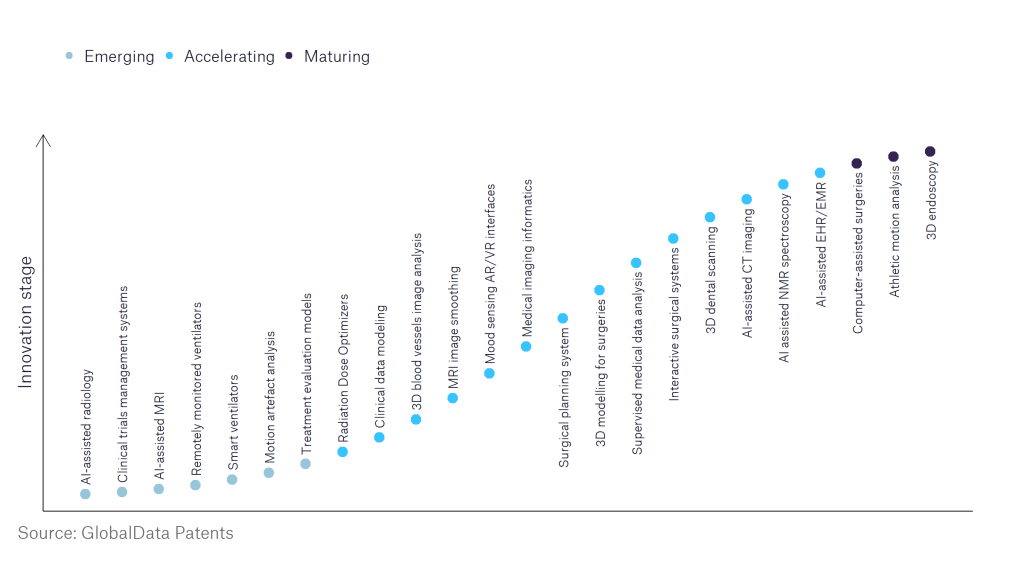
Regulation
A hurdle that AI technologies will face comes in the form of regulation. While government agencies scramble to understand and regulate this technology, it is constantly evolving.
“One of the biggest challenges in regulating AI in healthcare is the difference in pace between how long it takes for regulators to create and implement new policies compared to the time it takes software engineers and data scientists to create and update AI systems. Regulation can take years to enact, and we’ve all seen how fast the pace of AI advancement is where new capabilities are being announced weekly within the AI / technology sector,” said Sarah M. Worthy, CEO of DoorSpace a provider of a cloud space solution for clinicians.
She added: “We absolutely need stronger regulations around AI, and we cannot expect private, for-profit companies to self-regulate. I’m not sure how we can adapt our regulatory environment in this country in such a way as to protect consumers, but I do think it’s going to be the single most difficult challenge to regulating AI in medical devices.”
According to Murdoch, many medical device regulatory bodies don’t account for technologies that are changing in real time and many countries need to update their approval process to take this into consideration. “One of the biggest challenges when it comes to regulating AI in medical is the large datasets and complex algorithms used in the devices, so assessing functionality can be difficult without specific guidelines to follow,” said Murdoch.
AI imaging technology uses patients’ data, and this can become a concern when third parties’ business and insurers handle it as it could be vulnerable to hacking or missed used.
“Collaboration between AI developers, healthcare providers, and regulatory bodies is necessary to establish guidelines and standards for the development and deployment of AI-powered cancer imaging technologies. Ensuring transparency, explain ability, and accountability in AI algorithms is essential to build trust among healthcare professionals and patients,” said Murdoch.
The current picture
Artificial intelligence is revolutionising cancer imaging by enabling early detection, accurate diagnosis, personalised treatment, and improved workflow efficiency.
As technology continues to advance, AI-powered cancer imaging is poised to play a pivotal role in improving cancer detection rates, reducing false positives and false negatives, and ultimately saving lives. Embracing AI in cancer imaging requires a collaborative effort from researchers, healthcare professionals, and policymakers to overcome challenges and ensure its ethical and responsible use.
While the potential benefits of AI in cancer imaging are promising, it is essential to address the challenges and ethical considerations that arise. Additionally, maintaining patient privacy and data security is of utmost importance when handling sensitive medical information.





