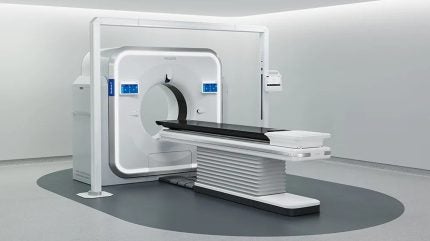
Philips has received the US Food and Drug Administration (FDA) 510(k) clearance for its Rembra platform of scanning systems, which covers the Rembra computed tomography (CT), Rembra radiation therapy (RT), and Areta RT systems.
These systems aim to support healthcare providers by improving speed, efficiency, and access to high-quality imaging as clinical and patient demands increase.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Designed to facilitate both diagnostic radiology and RT, the Rembra platform extends the capability of CT imaging to more connected workflows.
It also enables a coordinated approach to patient management, from rapid assessment in emergency settings to planning cancer treatments.
Philips CT business leader Dan Xu said: “As healthcare systems manage increasing demand and complexity, imaging plays a critical role in enabling timely and informed clinical decisions.
“With the Rembra platform, we are redefining what clinicians can expect from CT, combining speed, scalability, and precision to expand access to high-quality imaging while supporting confident diagnosis and highly accurate treatment planning.”
The Rembra CT system features an 85 cm bore and is designed to deliver ultra-fast, high-throughput imaging in intensive clinical settings, such as emergency departments and critical care.
It supports up to 270 exams per day, with advanced acquisition and reconstruction capabilities aimed at fostering timely decision-making without reducing diagnostic confidence.
Rembra RT and Areta RT systems are tailored for precision in radiation therapy. These platforms provide high-fidelity imaging for treatment planning, offering an 85 cm extended field of view and 4DCT imaging features intended to enhance tumour targeting and protect healthy tissue.
Their workflow streamlining is set to help decrease variability and optimise efficiency, supporting more tailored cancer therapies.
The Rembra CT imaging platform is intended to offer a unified high-performance CT ecosystem for urgent diagnosis through precision-guided therapy.
Philips aims to address expanding clinical needs with speed, consistency, and connection, while supporting both diagnosis and treatment workflows.
Earlier this year, Philips introduced the InkSpace Imaging Snuggle flexible paediatric body array coil, designed for use with its 3.0T magnetic resonance imaging (MRI) systems.



