FDA issues final rule regulating LDTs as medical devices
The FDA ruling follows years of industry back and forth and sees the government body choosing to exercise its right to impose regulations on the market.

The FDA ruling follows years of industry back and forth and sees the government body choosing to exercise its right to impose regulations on the market.


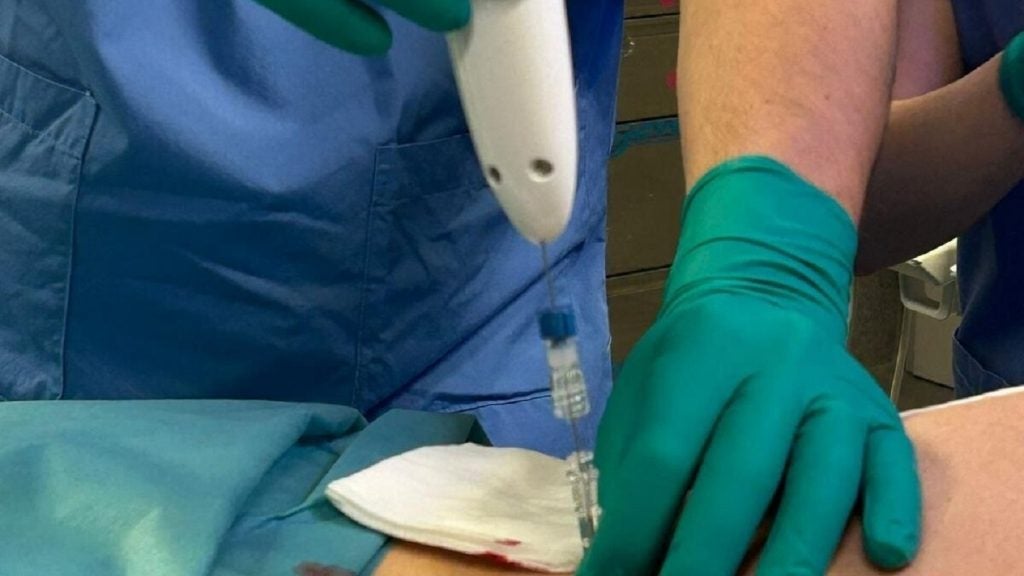
Dynamic Therapeutics’ U-Rhythm device captures hormone data over 24 hours and can help diagnose and monitor hormone-related disorders.
The medical devices industry continues to be a hotbed of patent innovation. Activity is driven by increased need for homecare,...
Revolutionising cardiac care: Caranx Medical makes history with robotic TAVI breakthrough
Hyperfine starts enrolment in portable imaging workflow study
Acarix starts clinical workflow study for its AI-powered cardiac diagnostic
MMI secures FDA approval for Symani Surgical System