In a recent article published by the BBC, it was found that a surprising fraction of the greenhouse gas emissions released by the NHS was from a single product: inhalers. Studies recommended switching some users from pressurised gas inhalers containing HFCs (potent greenhouse gases) to powder-based inhalers as it has been reported that this has a positive impact on both the planet and patients.
This change had the potential to deliver vast improvements to the carbon footprint of the NHS. But are there other significant improvements that can be made from alterations to medical products?
The medical devices field has been slow to change to sustainable practices in the way that other industries have. This is largely due to the higher levels of regulation, challenging requirements such as sterilisation and biocompatibility, and the need to undertake stringent re-validation and clinical evaluation if a significant change is made to an existing product.
Changes cannot be made at the expense of people’s health, hence risk and quality management standards are strict. However, regulations can constrain, impede or even discourage development and improvements, especially when sustainability improvements offer unclear financial or functional incentives.
Additionally, with public healthcare aiming to provide health coverage to as many people as possible, cost is an important factor to consider. To change a product, building a strong health economics case is essential.
When looking to make an improvement to your product’s sustainability, it is important to have a thorough understanding of its life-cycle and use. Understanding where the emissions are generated is key to making a meaningful impact when re-designing or optimising.
A study conducted by Fraunhofer UMSICHT showed that by using a Life Cycle Assessment (LCA), the associated emissions of a product could be halved by a change to the product’s use. The LCA compared multi-use catheters to single use and quantified the difference that change could make.
A full LCA conforming to ISO 14044:2006 can be a complex and expensive study. Whilst that will sometimes be the most appropriate option, we have found that adopting some LCA methodologies, without undergoing a full study, can help identify key areas of improvement quickly and cost-effectively.
The first step in using any LCA methodologies is defining the scope. This determines how much of the product journey will be examined, for example, if a product’s disposal or use will be considered. The flows of materials into and out of the different processes can then be estimated.

This investigation of the product life-cycle should identify the emissions sources and their scale. And when equipped with this knowledge, designers can assess which changes will have the biggest impact. Changing specific aspects of the product, its packaging materials or its geometry to increase the packing density during transport can then each be simulated in the LCA to establish the scale of the change and if there are any other repercussions.
Focusing on selected LCA methodologies, rather than investing in a full study, differs primarily in the level of detail that the study will go into. It uses general data sets and literature to provide an order of magnitude for different steps within the process and provides approximate relative contributions to the overall impact of a product.
Improving sustainability of packaging and disposables
One potential option for improving the sustainability of clinical products is reducing waste through redesign without impacting function.
Many surgical instruments are disposable for infection control reasons but companies such as Surgical Innovations are reducing the amount of material to be disposed of. Their Resposable products, for example, increase the modularity of clinical products to minimise the disposable components.
Another avenue of focus for the reduction of medical waste is the healthcare packaging industry which has historically focused on avoiding equipment contamination. Through careful material selection and design, companies such as Airnov and Amcor are releasing products designed to be recyclable without compromising functionality, thereby greatly reducing waste and associated emissions.
Improving sustainability of durable equipment
LCA techniques are even more powerful when assessing a product that has multiple cycles of use or a more complex life-cycle. Inhalers are a product that have an oft-repeated process which releases harmful gasses into the atmosphere throughout their life-cycle. Reusable or durable devices like this offer another opportunity for sustainability improvements. Unlike many disposables, the emissions are often not so heavily weighted in the manufacturing process but are more distributed throughout the use processes. Any improvement here could generate emissions savings many times over.
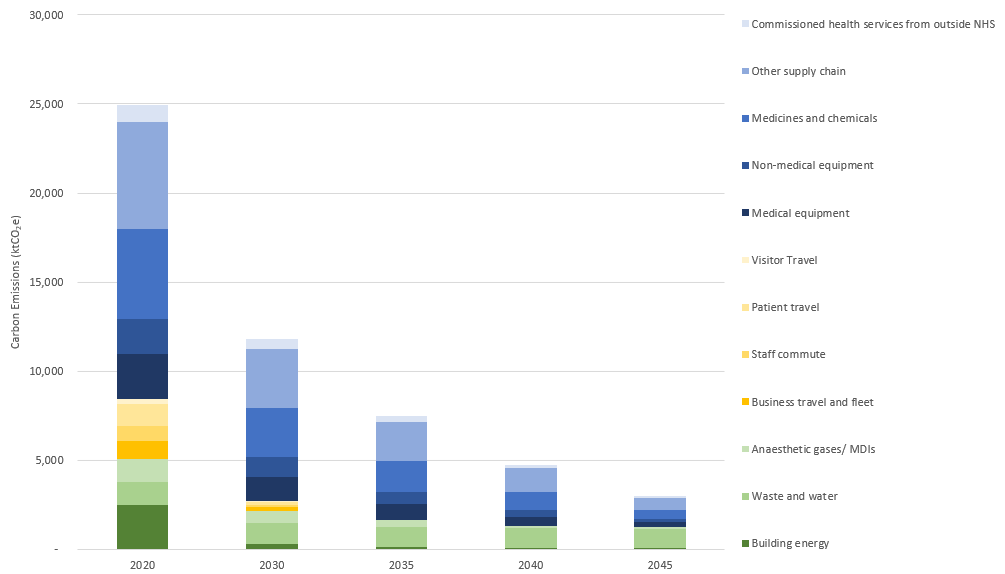
Reducing the energy consumption of hospital equipment would also have a great impact on the overall emissions of the NHS, as well as future technology and healthcare delivery innovations as shown in figure 2. As part of its net zero plan, published in July 2022, NHS England indicated that the design of new hospitals and replacement of existing equipment will be done “in a way that improves energy efficiency and reduces emissions”. As organisations worldwide commit to carbon reduction, energy efficiency will become a key selling point for new equipment.
A cost-effective first step
Our suggested process of using simplified LCA methodologies could be a cost-effective tool to understand and reduce your product emissions. Admittedly, the accuracy will be lower than that of a full study but for the reduced cost and time, these methodologies are a valuable tool.
Better understanding a product’s life-cycle can help you identify impactful potential alterations. These could involve minimal impact to design effort, clinical procedure, or patient experience, but still provide savings in carbon emissions that will help the sector make a more meaningful contribution towards net zero.
In addition, the UK government’s recently announced review of its net zero strategy (published 29 September) could lead to even greater incentives to invest in improving the sustainability of products within the medical device industry. But with the added requirements to be more pro-business and pro-growth while maintaining energy security, the government must recognise that supporting investment in developing products with lower carbon footprints, and improved energy efficiency, will be key.






