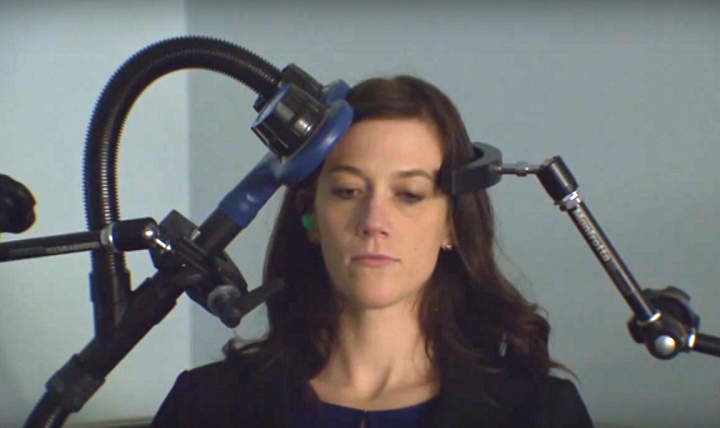
The US Food and Drug Administration (FDA) has permitted the marketing of the Brainsway Deep Transcranial Magnetic Stimulation System for treatment of obsessive compulsive disorder (OCD).
The Brainsway device is a non-invasive treatment that uses deep transcranial magnetic stimulation.
The treatment performs magnetic stimulation of frontal brain structures and networks, targeting previously unreachable nerve cells in the brain with its proprietary H7-coil, allowing it to effectively treat OCD.
Traits of obsessive compulsive disorder
OCD is characterised by the presence of obsessions or compulsions or both.
An obsession is defined as an unwanted intrusive thought, doubt, image or urge that repeatedly enters the mind.
Compulsions are repetitive behaviours or mental acts that a person feels driven to perform in response to an obsession.
OCD is the fourth most common mental disorder, with a lifetime prevalence of 1.6%.
The severity of the disorder can vary among individuals, and it can cause problems in relationships and health, as well as interfere with study or work.
OCD is ranked as one of the 10 most handicapping conditions by the World Health Organization.
Obsessive compulsive disorder treatment
Cognitive behavioural therapy (CBT) is an effective treatment for OCD.
Selective serotonin reuptake inhibitors (SSRIs) are the first-line drug treatment of choice because of better tolerability.
For patients for whom CBT and SSRIs have been ineffective, other treatments are available, such as ablative lesion neurosurgery, deep brain stimulation and transcranial magnetic stimulation.
“Transcranial magnetic stimulation has shown its potential to help patients suffering from depression and headaches,” said Carlos Peña, director of the Division of Neurological and Physical Medicine Devices in the FDA’s Center for Devices and Radiological Health.
“With today’s marketing authorisation, patients with OCD who have not responded to traditional treatments now have another option.”
For more insight and data, visit the GBI Research report store.