z-microsystems
Microfluidics and HQ Plastic Lenses
The success of the uneven but functional pairing of microfluidics and HQ plastic lenses has been confirmed in many specialised conferences and trade shows.
Subscribed
You have successfully submitted your enquiry. Someone from our company will respond ASAP
About Us
The success of the uneven but functional pairing of microfluidics and HQ plastic lenses has been confirmed in many specialised conferences and trade shows.
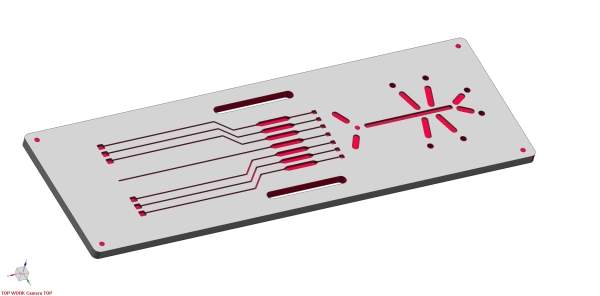
There is a current trend towards smaller and more complex structures. A significant factor here is the plastic lens technology within the field of microfluidics. They form the basis for analysis with laser units and require no assembly. z-microsystems is able to unite these two consumer requirements in one product and manufacture these lenses independently in an ISO-certified cleanroom.
These lenses can be used in a variety of applications; for example:
- Lab on a chip
- Microfluidics
- Microstructure
- Microreactor
- Microfilter
- Micropump
- Microvalve
- Microcomponents
- Microchannels
- Biochips
Special plastic injection molding technology
The ability to challenge forms in plastic injection molding is part of z-microsystems’ corporate philosophy and is reflected in more than 50 years of experience. The second image shown on the right shows a component which represents an enlarged view of the structure that can be produced in 1mm² on the basis of 76 holes with the square dimensions of 40μ each. z-microsystems uses special mold and injection molding technology with plastic piles which have a holding or positioning function. The challenge here is to operate efficiently and without the use of deposits or additional complexity.
Plastic optical lenses for the medical industry
Plastic optical lenses are not only used in the defined areas of operation (microscopes, fibre optics, etc.), but also in the areas of analysis and diagnosis in the medical industry. Glass lenses, which were assembled by additional assembly processes, are being replaced with plastic lenses more and more. The reason for this is that there are no losses in the analysis process compared to glass lenses.
Plastic injection molding – full development and production support
The above described areas in plastic injection molding applications are just part of our manufacturing capabilities. We provide our customers with technical support for their products during the development process. This begins with on-site support of the first developing parts for internal laboratory tests, followed by pre-series as well as series production.
With ISO 9001 and ISO 13485 medical devices, we take account of market demand. Ask us for further information and see how our experience and manufacturing expertise can benefit you.
Contact Details
Website
Email Address
Address
6850 Dornbirn,
Austria