Medical Device Manufacturing and Engineering
TRICOR Systems Inc, winner of the 2008 Medical Design Excellence Award, is an FDA registered, ISO 9001:2015 and ISO 13485:2016 medical manufacturer and design facility.

TRICOR Systems provides reliable high-quality electronic contract manufacturing services for medical device companies.
You have successfully submitted your enquiry. Someone from our company will respond ASAP

TRICOR Systems provides reliable high-quality electronic contract manufacturing services for medical device companies.
Our award-winning patentable products highlight our reputation for innovative design and quality manufacturing.
Our engineers’ knowledge of optics, mechanics and electronics allow for unique design for manufacturability support for specialised and unique equipment within the electronic contract manufacturing medical market.
TRICOR Systems is an ISO 13485: 2016 certified, Food and Drug Administration (FDA) registered medical manufacturing facility. Established in 1976, we provide high-quality electronic contract manufacturing to the medical industry.
Our employees are skilled in manufacturing Class I, II and III electronic medical devices, starting from supplementation of internal capabilities to complete turnkey solutions. We have the experience and flexibility to meet customer requirements, providing a high-quality reliable product.
TRICOR is familiar with the ebb and flow of start-up companies manufacturing requirements and work with them to meet their needs. TRICOR offers a wide variety of services ranging from short-run prototypes to full production. TRICOR excels at complete, fully tested box builds, cables, harnesses, circuit card assemblies and build to print. Custom electronic equipment is our speciality.
The company has established a reputation for its high quality and on-time delivery.
TRICOR has developed Dri-Scope Aid Cabinet and The Dri-Scope Aid ~ Jet Stream to aid in drying the internal channels of an endoscope.
The device helps to prevent microbiological growth, thus preventing post-operative infection.
We have spent several years producing the LKT100P and LKT101P Kidney Transporter Devices and have successfully design and develop the LifePort liver transport (LTT) solution.
Similarly, to the LKT, the LLT uses a peristaltic pump to support the liver during transport, providing a more sustainable transplant organ.
TRICOR Systems designs the electrical components and establishes the software for medical award-winning STA, a single tooth computer-controlled local anaesthesia delivery system.
We are currently manufacturing the device, which provides almost completely pain-free and fear-free injections. We also help to write technical segments of FDA 510K submissions for such Class II devices.
The CompuFlo unit is a computer-controlled anaesthesia delivery system for epidurals. Its prototype has been developed to support doctors to administer a safer epidural spinal injection.
We also assist in writing technical segments of FDA 510K proposals for this type of Class II device, with an aim for approval on the first submission.
Since 2006, TRICOR has been manufacturing the Class II device, the first automated disinfector designed to offer high-level disinfection of transesophageal (TEE) ultrasound probes.
The microprocessor-controlled device offers simple interaction by the healthcare professional responsible for reprocessing the used ultrasound probe.
TRICOR manufactures the endoscope leak detection system, a Class II device that detects leaks with almost 100% accuracy.
It identifies endoscope leaks by using an air pump to pressurise the endoscope, as well as monitor fluctuations in pressure. Early leak detection significantly reduces repair costs and the risk of cross-contamination.
We had been commissioned to manufacture and subsequently re-engineer the TMX 2000 to the TMX 3000.
The TMX 3000 is a Class III device to treat the prostate and an updated version of the TMX 2000, which went end-of-life after being replaced by alternative technology.
TRICOR is an ITAR and FDA-registered company, which maintains AS9100D, ISO 9001: 2015 and ISO 13485: 2016 certifications.
We function as an electronic contract manufacturer of stock and custom instrumentation products for start-up to fortune 500 companies.
Our knowledge involves the engineering of electronic systems, electronic contract manufacturing, cables, circuit cards and electro-optical mechanical devices for medical, military, industrial and aerospace operations.

TRICOR Systems Inc, winner of the 2008 Medical Design Excellence Award, is an FDA registered, ISO 9001:2015 and ISO 13485:2016 medical manufacturer and design facility.

Convert your existing scope cabinet into an automatic scope drying system.

The TRICOR Model 807A DOI/Haze Meter offers rapid, precise measurement of gloss, distinctness of image, and haze on high reflectance surfaces, enabling operators to easily differentiate subtle variations in reflectance characteristics with just a single button press.
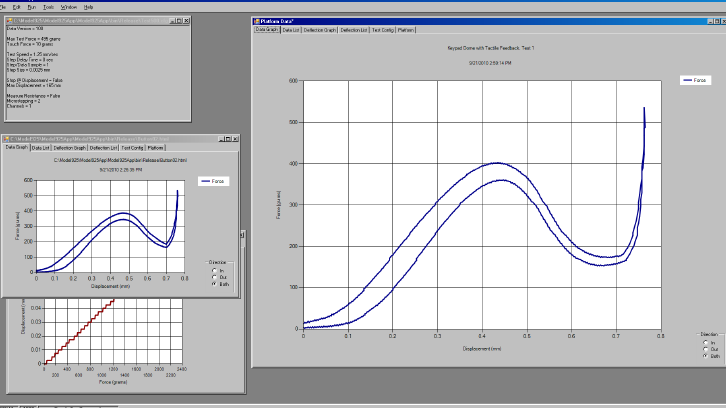
The Model 925 Force-Distance Tester is an affordable, high-precision testing solution that seamlessly integrates with Windows™ PCs to deliver accurate force and displacement data in Excel format, enabling efficient analysis and reporting for quality assurance, engineering investigations, and life studies.
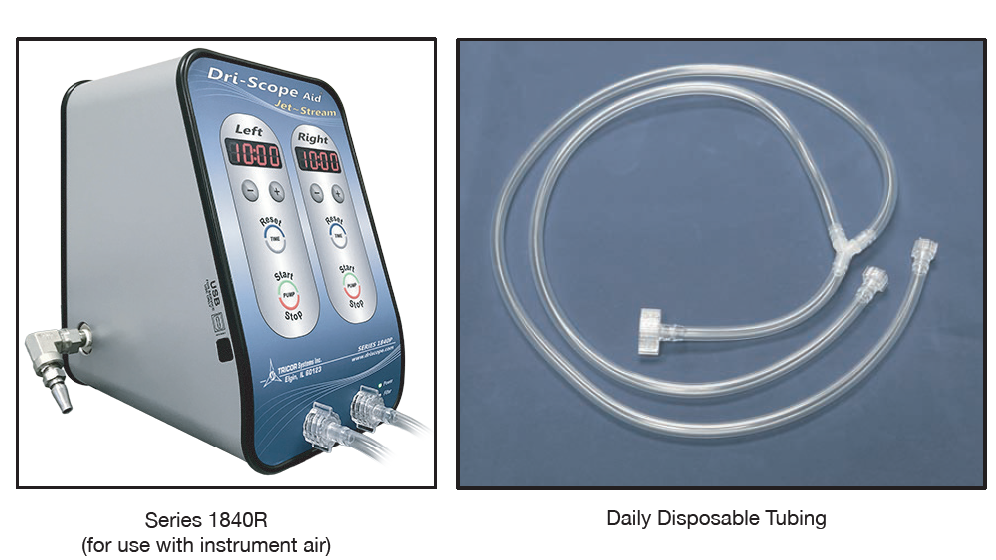
The Dri-Scope Aid® Jet~Stream is a versatile, hands-free drying solution designed to efficiently purge and dry the internal channels of endoscopes with HEPA-filtered air, featuring dual timers for simultaneous operation of two scopes, audible and visual alerts, and easy mounting options for enhanced convenience in medical settings.

Introducing the TD-702 REV B brochure, your comprehensive guide to market-leading dryers for internal endoscope channels, designed to ensure compliance with the latest AAMI, AORN, and SGNA guidelines while enhancing workspace efficiency and drying capabilities with innovative features like the Jet~Cart and Jet~Pole systems, as well as the Dri-Scope Aid Cabinet for seamless integration into existing setups.