Laboratorios Farmaceuticos ROVI
Seeking Partners for Fibrilin, a Catheter Lock Solution
Laboratorios Farmaceuticos ROVI is a Spanish speciality pharmaceutical company that has been manufacturing and marketing the medical device Fibrilin® in Spain and Portugal for more than ten years.
Subscribed
You have successfully submitted your enquiry. Someone from our company will respond ASAP
About Us
Laboratorios Farmaceuticos ROVI is a Spanish speciality pharmaceutical company that has been manufacturing and marketing the medical device Fibrilin® in Spain and Portugal for more than ten years. The company is currently seeking partners to bring Fibrilin to new markets.
Our in-house R&D core experience is in the field of heparin. We are fully integrated from manufacturing API to filling, packaging, marketing and distribution. Our portfolio includes Bemiparin (low molecular weight heparin, marketed in 52 countries through exclusive domestic distributors), heparin sodium (approved in Spain and updating dossier for EU countries) and Fibrilin, which is available in EU countries and countries where the CE mark is accepted.
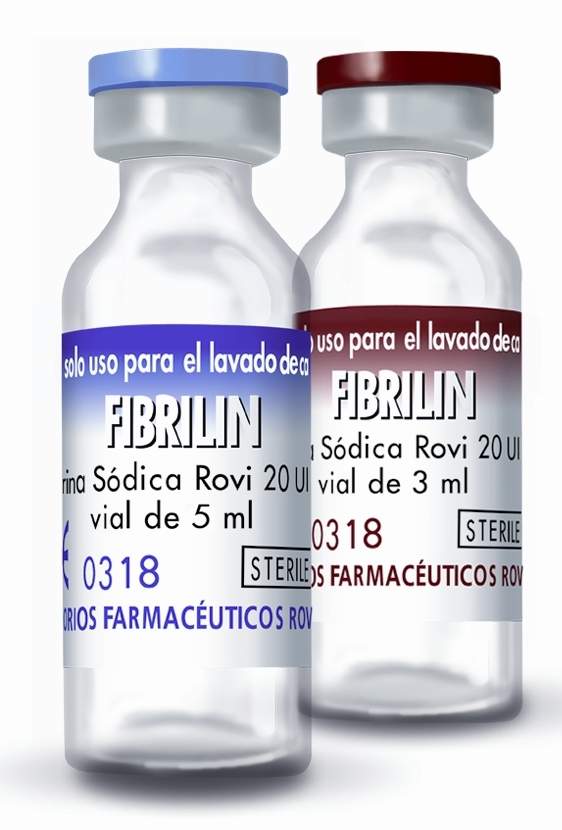
Fibrilin sodium heparin solution in unit dose vials
Fibrilin is the innovative catheter lock solution for nurses to prevent fibrin formation in both central and peripheral venous catheters and its consequences (phlebitis, infections, etc.) for hospitalised patients. It increases quality of life and reduces costs, with the minimum workload among available lock solutions. This is due to Fibrilin’s ability apply positive pressure to the blood stream.
Effective and financially competitive, Fibrilin is the best solution compared to classic heparin sodium dilution multidose.
Ready-to-market catheter lock solution in the EU
Fibrilin is ready to market in EU countries and countries where the CE mark is accepted. The solution is currently marketed in Spain and Portugal, where we market the product ourselves.
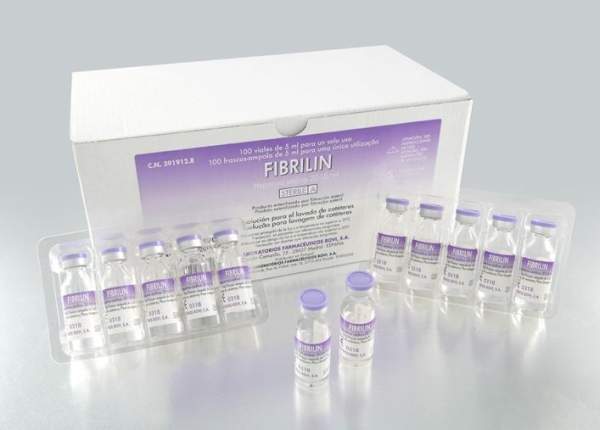
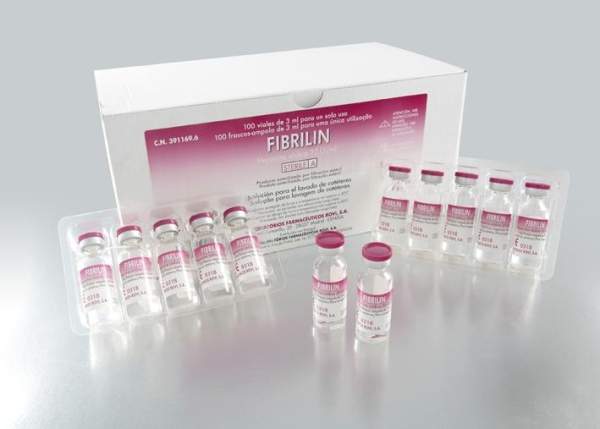
As Fibrilin does not present therapeutic activity, it is registered as medical device class III – CE mark. It is available in two presentations with sterile unit dose vials:
- Fibrilin 3ml – 100 vials/box (60 IU/vial)
- Fibrilin 5ml – 100 vials/box (100 IU/vial)
Maintaining catheter patency
Fibrilin maintains catheter patency, avoiding fibrin formation and its consequences, including phlebitis, nosocomial infections, introduction of new catheters, thrombosis, extravasation and perfusion / aspiration.
There are a number of benefits to the solution. Fibrilin has the following benefits:
- Improves quality of life for patients
- Decreases health sanitary cost
- Reduces nurses’ workload
Lock solution for peripheral venous and central venous catheters
Both peripheral venous catheters and central venous catheters should be locked with Fibrilin by nursing staff after each use and when the catheter has an intermittent use. Therefore, Fibrilin should be present in all departments of each hospital. Maintaining patency is essential for central venous catheters. For this reason, we suggest the following departments as places for Fibrilin to be stocked:
- Nephrology (haemodialysis)
- Oncology
- Haematology and haemotherapy
- Cardiovascular surgery
- Internal medicine
- Intensive care
Medical device marketing opportunities
ROVI is seeking a partner that could position our product in an efficient way, and would hold the exclusive rights of Fibrilin in each country where Fibrilin could be marketed, except Spain and Portugal.
About Laboratorios Farmaceuticos ROVI
Laboratorios Farmaceuticos ROVI is fully-integrated, profitable, Spanish speciality pharmaceutical company engaged in the research, development, manufacturing and marketing of small molecule and speciality biologic drugs. It was founded in 1946 and has been quoted on the Madrid Stock Exchange since 2007. 2011 revenues were €184.7m, and we have over 920 employees.
If you are interested in our product or becoming a partner, please do not hesitate to contact us by phone, email or via the form on this page.
Contact Details
Website
Email Address
Address
28037 Madrid,
Spain