GM Nameplate
Custom and Fully Assembled User Interfaces
Subscribed
You have successfully submitted your enquiry. Someone from our company will respond ASAP
About Us
GM Nameplate is an industrial manufacturer that provides medical OEMs with custom user interface designs, single-use biosensors, diagnostics test strips, as well as fully assembled user interfaces including graphic overlays, membrane switches, rubber keypads, touch screens, injection and compression molding and plastic decorating, shielding and other die-cut components.
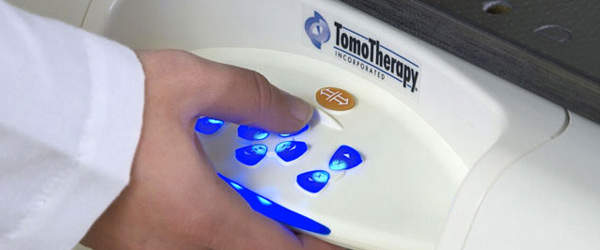
Medical device user interface components
We integrate multiple technologies and understand that building user interface components and working with one vendor instead of several saves you both time and money. As a result, we provide you with a full package solution.
Working with our customers in the development of their new product platforms in relation to user interface designs, we offer multiple technologies to create the most efficient and cost-effective solutions in meeting and exceeding their product specifications. We are an industry leader in the user interface, bio-sensor / electrode arena, along with printing in the medical field through such industries as portable DNA labs, diabetic test strips and iontophoresis patches.
It is our mission to produce products that precisely meet customer expectations on time, every time, at a projected or reduced cost. We provide product development assistance and a broad range of manufacturing capabilities to take your project from concept to conclusion.
User interface component manufacturing and assembly
GM Nameplate prints, fabricates and assembles the various user interface layers: graphic overlay, touch screen, membrane, shielding, spacer, subpanel and housing. Diverse manufacturing capabilities and the upfront design assistance of our experienced engineering staff ensure that your product fulfills functional requirements and maximizes the ergonomic potential of your design. When you need a quick-turn membrane switch prototype, GM Nameplate can produce it in ten business days or less (depending on design complexity).
Biosensor / diagnostic test strip printing and converting
Biosensor development requires coordinating many unique resources. Each sensor uses specially formulated substrates and chemicals and requires carefully devised printing, converting and manufacturing techniques. GM Nameplate’s range of capabilities, extensive printing and converting experience, access to cutting edge materials and technologies and dedication to quality and R&D make our company a trusted manufacturing source for many of today’s largest medical companies in the biosensor market.
Force-sensitive resistors
GM Nameplate provides force-sensitive resistors that react to pressure and can accept multiple concurrent inputs. This feature enables surface area, time and weight estimations not obtainable from standard input options such as membrane switches or touch screens. Our force-sensitive resistor technology allows the creation of custom sizes and shapes. If your application requires determining input involving pressure / weight, duration and surface area of a touch, force-sensitive resistors are a viable option for you.
Optical bonding and liquid optical bonding
GM Nameplate’s optically bonded technology provides our customers with the very latest in display enhancement capabilities. This is a high-performance adhesive and proven bonding process that optimizes the view-ability and durability of LCD displays. When used in the construction of front panels during manufacturing, the optical bonding technology significantly increases LCD readability in bright sunlight while also providing dramatically improved scratch resistance, impact resistance and overall durability to moisture and dust.
Liquid optical bonding combines the benefits of silicone and epoxy while remaining optically clear and highly resistant to yellowing, delamination and bubbling.
Global medical device manufacturing solutions
Producing quality components that exceed your expectations is our ultimate goal. All of GM Nameplate’s eight facilities located across North America and Asia practice Current Good Manufacturing Practices (cGMP) and are ISO 9001 and ISO 14001:2004 approved.
Contact us to learn more about how our custom manufacturing experience and expertise can complement your internal resources and enable you to launch products to market faster.
White Papers
Contact Details
Website
Email Address
Address
WA 98119,
Seattle,
Other,
United States of America