Clearside BioMedical has filed a patent for a non-surgical method of treating ocular disorders by administering gene therapies to the suprachoroidal space of the eye. The method aims to achieve expression of the transgene in the posterior segment of the eye. GlobalData’s report on Clearside BioMedical gives a 360-degree view of the company including its patenting strategy. Buy the report here.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
According to GlobalData’s company profile on Clearside BioMedical, wearable drug delivery devices was a key innovation area identified from patents. Clearside BioMedical's grant share as of June 2023 was 1%. Grant share is based on the ratio of number of grants to total number of patents.
Non-surgical gene therapy for treating ocular disorders
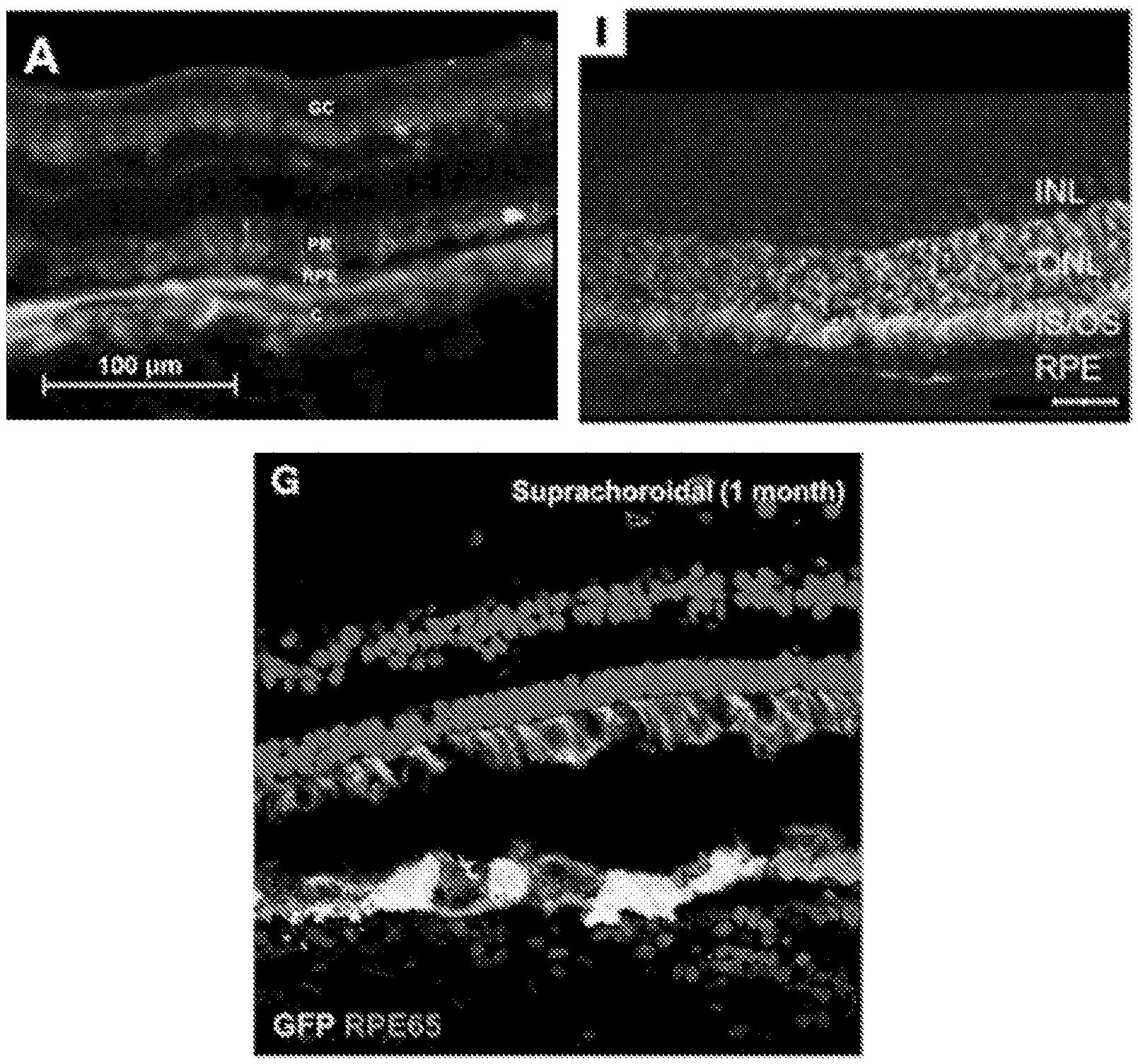
A recently filed patent (Publication Number: US20230201371A1) describes a method for treating ocular disorders non-surgically. The method involves administering a vector containing a transgene to the suprachoroidal space (SCS) of the eye, resulting in the expression of the transgene in the posterior segment of the eye. The vector can be a viral vector, such as AAV8, or a non-viral nanoparticle. The method may also include administering a steroid or an anti-inflammatory agent to the eye. Additionally, the intraocular pressure of the eye can be modulated before or after vector administration. The ocular disorders that can be treated using this method include diabetic retinopathy and neovascular age-related macular degeneration (nAMD). The transgene can encode a therapeutic agent, such as a cytokine, growth factor, anti-angiogenesis factor, or an antibody. The vector can be administered to the SCS using a hollow microneedle. The method allows for targeted treatment of ocular disorders and has the potential to be effective in humans.
Another aspect of the patent describes a method for treating ocular disorders by administering a therapeutic agent to the suprachoroidal space (SCS) of the eye. This method involves various techniques to enhance the effectiveness of the treatment. These techniques include expanding the SCS before administering the therapeutic agent, manipulating the intraocular pressure, administering a pushing formulation or a steroid, injecting the therapeutic agent in multiple quadrants of the eye, modulating the speed of injection, applying iontophoresis, using high viscosity or hyaluronic acid formulations, conjugating the therapeutic agent to a large polymer, injecting a specific volume or high dose, and using a highly soluble formulation. These techniques aim to optimize the delivery and distribution of the therapeutic agent within the posterior segment of the eye. The method provides a comprehensive approach to treating ocular disorders and offers potential benefits for patients in need of effective treatment options.
In summary, the filed patent describes a non-surgical method for treating ocular disorders by administering a vector or therapeutic agent to the suprachoroidal space of the eye. The method offers targeted treatment and potential benefits for patients with ocular disorders, including diabetic retinopathy and neovascular age-related macular degeneration. Various techniques are proposed to enhance the effectiveness of the treatment, such as modulating intraocular pressure and using specific formulations and delivery methods. The patent has the potential to advance the field of ocular therapeutics and improve patient outcomes.
To know more about GlobalData’s detailed insights on Clearside BioMedical, buy the report here.
Data Insights
From

The gold standard of business intelligence.
Blending expert knowledge with cutting-edge technology, GlobalData’s unrivalled proprietary data will enable you to decode what’s happening in your market. You can make better informed decisions and gain a future-proof advantage over your competitors.




