Daxor has filed a patent for a system and method to measure blood volume using a fluorescent dye. The invention simplifies the process by using a fluorescent tracer and includes an injection and sampling kit, as well as a calibration kit. The system automatically analyzes blood samples, measures fluorescence levels, and calculates blood volume, plasma volume, and red cell volume for a living subject based on known concentrations of the fluorescent tracer. The results are displayed on a user interface. GlobalData’s report on Daxor gives a 360-degree view of the company including its patenting strategy. Buy the report here.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
According to GlobalData’s company profile on Daxor, sleep quality monitoring was a key innovation area identified from patents. Daxor's grant share as of June 2023 was 1%. Grant share is based on the ratio of number of grants to total number of patents.
Blood volume measurement using fluorescent dye and calibration cassettes
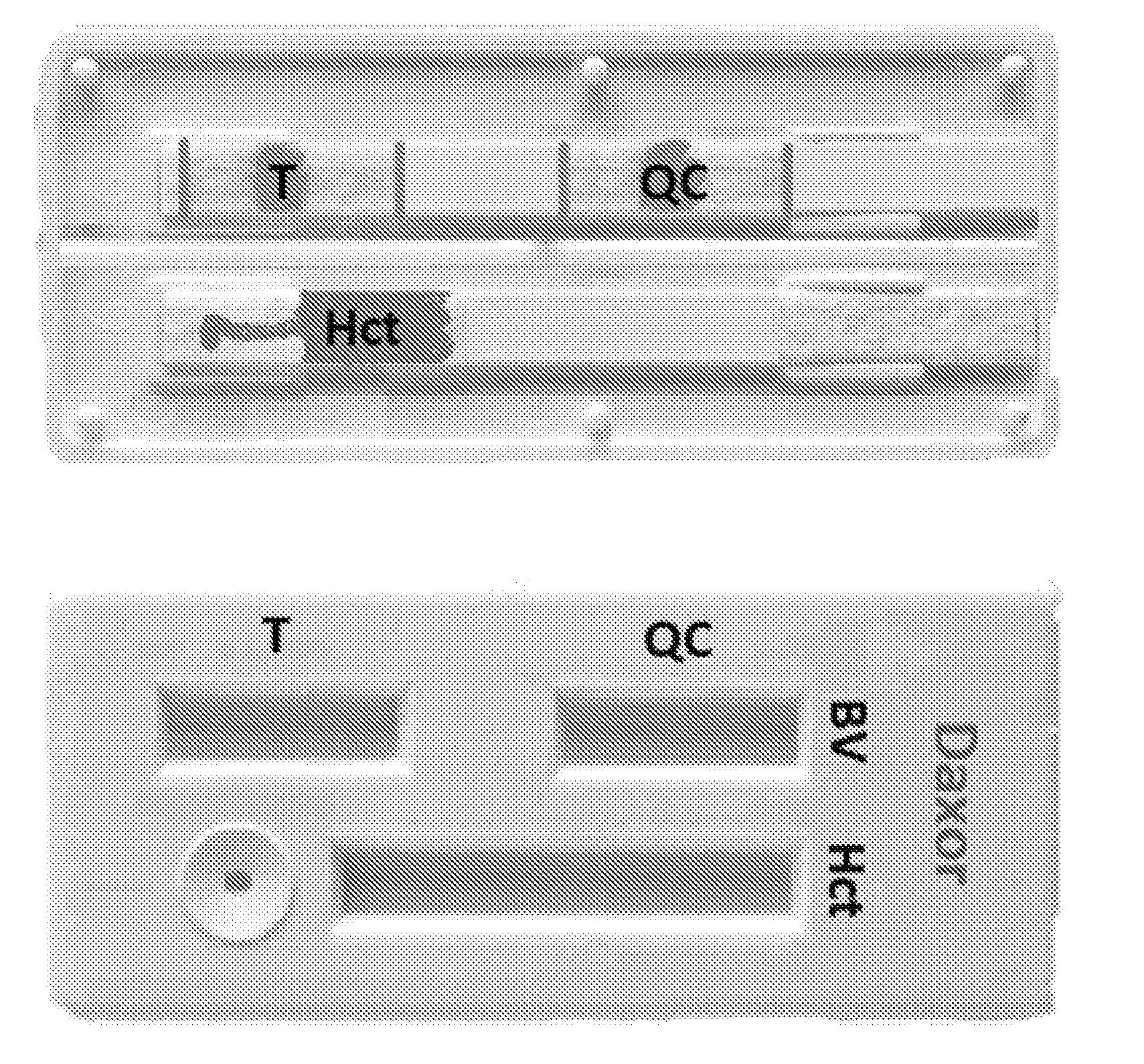
A recently filed patent (Publication Number: US20230131712A1) describes a system for automatically analyzing blood in a living subject. The system includes multiple concentration-measuring cassettes, calibration cassettes with known concentrations of fluorescent activity, a reader for measuring fluorescence levels, and a user interface for input and display of information. The system also includes processors and memory to execute programmed instructions for carrying out the analysis.
The method described in the patent involves several steps. First, a sample of whole blood is measured in a counting cassette to determine the background level of fluorescence. Then, the subject is injected with a precise, known volume of fluorescent tracer. The level of fluorescence is measured in calibration cassettes matched to the batch of tracer used. A calibration curve is created to relate fluorescence to volume of dilution. At timed intervals after the injection, samples of whole blood are placed in counting cassettes and the post-injection level of fluorescence is measured. The volume of dilution for each sample is quantified using interpolation between the measured activity of the calibration cassettes and their known concentrations. The processors then calculate the blood volume, plasma volume, and red cell volume for the subject based on this information. Additionally, an ideal blood volume, ideal plasma volume, and red cell volume can be calculated based on subject descriptive data such as height, weight, and gender. The results are displayed at the user interface.
The patent also describes various features of the system, such as the use of separate receptacles in the sample collection cassettes for determining Hematocrit (Hct), the use of membrane-based cassettes with full-wicking or lateral flow methodology, and the use of an integrated device with a touchscreen and barcode reader for input and measurement of cassettes.
Furthermore, the patent includes claims for an injection and sampling kit, a calibration kit, and a full measurement kit for performing the indicator dilution measurement. The method described in the patent can be used with these kits to determine the blood volume of a subject. The counting cassettes may also contain an aperture for measuring Hct, and the method can be performed using membrane-based cassettes with full-wicking or lateral flow methodology.
To know more about GlobalData’s detailed insights on Daxor, buy the report here.
Data Insights
From

The gold standard of business intelligence.
Blending expert knowledge with cutting-edge technology, GlobalData’s unrivalled proprietary data will enable you to decode what’s happening in your market. You can make better informed decisions and gain a future-proof advantage over your competitors.




