The medical devices industry continues to be a hotbed of innovation, with activity driven by increased need for homecare, preventative treatments, early diagnosis, reducing patient recovery times and improving outcomes, as well as a growing importance of technologies such as machine learning, augmented reality, 5G and digitalisation. In the last three years alone, there have been over 450,000 patents filed and granted in the medical devices industry, according to GlobalData’s report on Internet of things in Medical Devices: Programmable infusion pumps. Buy the report here.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
However, not all innovations are equal and nor do they follow a constant upward trend. Instead, their evolution takes the form of an S-shaped curve that reflects their typical lifecycle from early emergence to accelerating adoption, before finally stabilising and reaching maturity.
Identifying where a particular innovation is on this journey, especially those that are in the emerging and accelerating stages, is essential for understanding their current level of adoption and the likely future trajectory and impact they will have.
150+ innovations will shape the medical devices industry
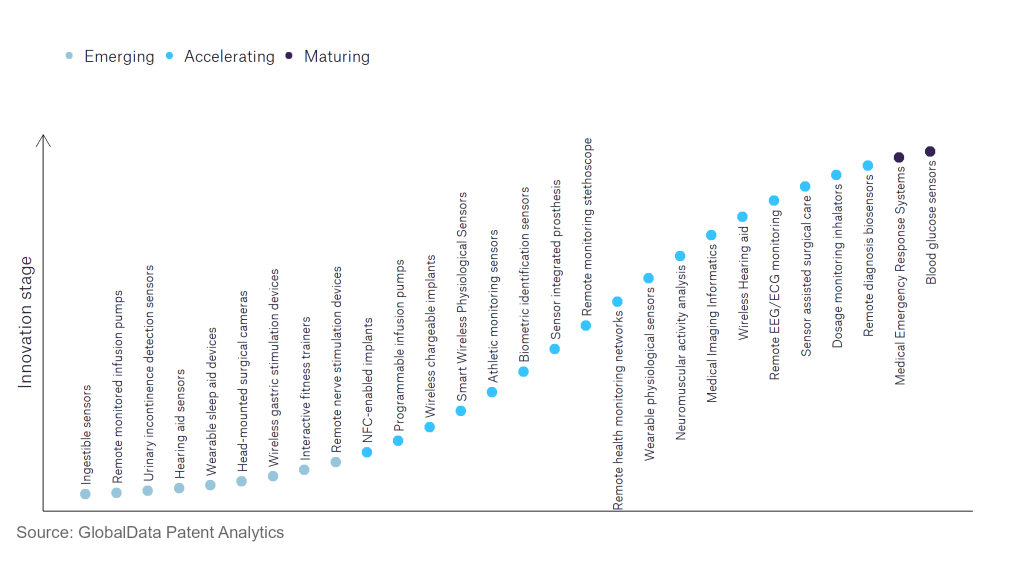
According to GlobalData’s Technology Foresights, which plots the S-curve for the medical devices industry using innovation intensity models built on over 550,000 patents, there are 150+ innovation areas that will shape the future of the industry.
Within the emerging innovation stage, ingestible sensors, wireless gastric stimulation devices, and remote nerve stimulation devices are disruptive technologies that are in the early stages of application and should be tracked closely. dosage monitoring inhalators, programmable infusion pumps, and athletic monitoring sensors are some of the accelerating innovation areas, where adoption has been steadily increasing. Among maturing innovation areas are blood glucose sensors and medical emergency response systems, which are now well established in the industry.
Innovation S-curve for Internet of Things in the medical devices industry
Programmable infusion pumps is a key innovation area in Internet of Things
A programmed infusion pump, also known as a smart pump, is a device that comprises a drug library and is also known as a dosage error-reduction system. Smart pumps assist the administration of intravenous fluids and drugs while adhering to predefined parameters such as drug concentration and dosage.
Infusion pumps are typically operated by a skilled user who programmes the rate and duration of fluid delivery using a built-in software interface. Pump failures can have serious consequences for patient safety since infusion pumps are regularly used to give vital fluids, including high-risk drugs. Many infusion pumps have safety measures such as alarms or other operator alerts that are designed to trigger in the case of a malfunction.
GlobalData’s analysis also uncovers the companies at the forefront of each innovation area and assesses the potential reach and impact of their patenting activity across different applications and geographies. According to GlobalData, there are 30+ companies, spanning technology vendors, established medical devices companies, and up-and-coming start-ups engaged in the development and application of programmable infusion pumps.
Key players in programmable infusion pumps– a disruptive innovation in the medical devices industry
‘Application diversity’ measures the number of different applications identified for each relevant patent and broadly splits companies into either ‘niche’ or ‘diversified’ innovators.
‘Geographic reach’ refers to the number of different countries each relevant patent is registered in and reflects the breadth of geographic application intended, ranging from ‘global’ to ‘local’.
DEKA Research and Development is the leading patent filer in programmable infusion pumps market. Some other leading patent filers include Sanofi, Medtronic and F. Hoffmann-La Roche.
In terms of application diversity, Bristol-Myers Squibb leads the pack, followed by Baxter International and West Pharmaceutical Services. With regards to geographic reach, Platinum Equity leads, followed by B. Braun Miethke and Bristol-Myers Squibb.
Significant safety concerns about infusion pumps have been brought to the FDA's notice in recent years. These problems can risk the safe use of external infusion pumps, resulting in over- or under-infusion, missed treatments, or delayed medication. As with any technology, new smart-pump capabilities have potential obstacles to overcome. Integration with laboratory data would need to leverage novel software. Monitoring and titration procedures would be automated, reducing nurses' opportunity to develop these abilities. Finally, new safety systems have the potential to develop new faults. As a result, growing smart-pump applications will demand cautious development and testing.
To further understand the key themes and technologies disrupting the medical devices industry, access GlobalData’s latest thematic research report on Medical Devices.
Data Insights
From

The gold standard of business intelligence.
Blending expert knowledge with cutting-edge technology, GlobalData’s unrivalled proprietary data will enable you to decode what’s happening in your market. You can make better informed decisions and gain a future-proof advantage over your competitors.




