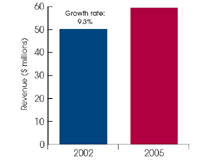
In 2005, the European bone graft market was estimated by Frost &
Sullivan to be worth close to $60m. It is a field that has seen considerable
growth recently. One reason for this is the ageing European population, which
has experienced a rise in age-related musculoskeletal disorders such as
osteoporosis and osteoarthritis.
Another crucial factor contributing to market growth is smoking, which has
an effect on bone density and so influences the occurrence of bone disease.
However, the number of smokers in Europe as a percentage of population has been
decreasing in recent years. As a result, the effects of smoking on the bone
graft market are expected to start diminishing in the future.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Supplies of allografts from cadavers are limited and cannot always meet
demand, and some other forms of graft, such as hydroxyapatite and demineralised
bone matrix (DBM), are made from allografts. These factors are driving a move
towards bovine bone and synthetic bone grafts.
NEW REGULATIONS FOR BOVINE GRAFTS
In 2004, EU directive 2003/32/EC came into effect, requiring all
manufacturers to undergo additional regulatory assessment procedures before
allowing the introduction of any products using material of animal origin to
the EU market. The new regulations, introduced primarily to minimise the
substantial risk of spreading transmissible spongiform encephalopathies (TSEs),
have been made applicable to all existing and new products.
For bone grafts, the risk of TSE transmission can be minimised by sourcing
tissue from countries free from bovine spongiform encephalopathy (BSE), such as
Australia, New Zealand, Argentina and Brazil. The alternative – putting
tissue through a very harsh process to eliminate disease-carrying prions
– severely affects the mechanical properties of the material and usually
means that it is unsuitable for use as a bone graft.
The enhanced control required by these regulations has had an impact on the
price of bone grafts, as well as their availability. There are now fewer
manufacturers working in this field because of the extra costs of production.
However, bovine bone grafts are still more readily available than allografts
and, with the extra controls, should pose even less risk in terms of
transmitting disease to patients.
PRODUCT DEVELOPMENT AND NEW TRENDS
Manufacturers are increasingly offering new materials and combinations of
existing materials for bone replacement In general, no bone graft substitute is
as good as an autograft. It is only in situations where the patient cannot
undergo an autograft that an alternative is likely to be used. In this case, if
the bone graft is required to provide mechanical strength, bovine bone grafts
are the best option.
Bone grafts made of a combination of bovine collagen and hydroxyapatite
provide mechanical strength and induce new bone growth. If the patient does not
require mechanical support, bioglass offers better results than the more common
hydroxyapatite or DBM.
Since the introduction of bone grafts, surgeons have gradually started using
them instead of the traditional metal plates, pins and screws. However, the
limited clinical application of bone grafts in situations where an allograft
cannot be used has meant that many doctors experienced in using metal implants
do not see the need to learn a new technique for a limited number of
patients.
To change the practice of these surgeons, more positive clinical data
demonstrating the advantages of bone grafts over metal implants are required.
The fact that bone grafts prevent the need for a second surgical site, and do
not require surgery to remove the implant once the bone has healed, has been a
considerable factor in ensuring the widespread uptake of these devices in
recent years.
PROMISING MARKET OUTLOOK
One restraint to further growth of the market is the increasing budgetary
constraint faced by European healthcare providers. Manufactured bone grafts are
more expensive than autografts (which are freely donated by the patient) and
also tend to be more expensive than processed allografts.
However, Frost & Sullivan predicts that the European bone graft market
will continue to grow in the coming years. This is due again to an ageing
population increasing the number of patients requiring manufactured bone
grafts, alongside greater acceptance of these products among clinicians.
While recent regulatory changes have restrained the market, they will ensure
that bone grafts used are safe for the patient. This, in turn, should guarantee
continued confidence in the use of non-human bone grafts.





