For over 20 years, efforts have been made to standardise and harmonise electronic network technologies for medical applications. These efforts have yielded the well-known DICOM network for image processing, the Health Level Seven (HL7) initiative to standardise hospital backbone networks and the ISO 11073 medical information bus (also known as IEEE 1073) for intensive care units and operating rooms. Meanwhile, manufacturers of medical devices have developed many proprietary network solutions that are embedded in their medical devices.
CAN AND CANopen

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
As medical devices become more sophisticated, manufacturers are no longer developing and producing all the parts themselves. To reduce development costs and meet time-to-market requirements, they buy off-the-shelf products to integrate into their medical devices. As a consequence, open interface standards are important to medical device manufacturers, not just because they simplify the integration of third-party products, but also because they allow the substitution of one product with an alternative from another manufacturer.
More than ten years ago, several manufacturers of medical devices and healthcare equipment started to use the serial Controller Area Network (CAN) bus system as an embedded network for real-time control.
CAN was originally developed for the automotive industry and is one of the most reliable communication networks. The internationally standardised network (ISO 11898-1/2) features many built-in error detection mechanisms, as well as sophisticated fault confinement.
All major semiconductor manufacturers provide a micro-controller with on-chip CAN controllers. However, the CAN protocol covers only the lower network layers, such as telephone lines. The higher layer protocols comparable with human language are not standardised by the CAN specifications.
About ten years ago, the EU initiated a research project to develop a CAN based higher-layer protocol and device profiles for embedded control networks. The results of this project were passed on to the CAN in Automation (CiA) association.
Meanwhile, this application layer and communication profile has been internationally standardised (EN 50325-4), and is known as CANopen. CANopen networks are used in many different application fields, including medical devices and healthcare equipment.
There are several benefits to be gained by using standardised networks such as CANopen. For example, CANopen software stacks can be purchased from different suppliers. Third-party CANopen tools for development, configuration and diagnostics are available. Training on the software, protocol or tools is available from outside companies, which frees up internal resources.
STANDARDISED DEVICE PROFILE
According to Don Medlar from Siemens Medical Solutions: “With proprietary protocols, component manufacturers found themselves faced with the problem communication protocols, even though basically the same functionality was being used.”
To prevent this from happening all the time, CiA has developed several dedicated device profiles for medical devices and healthcare equipment. Such device profiles specify exactly the process data, and the application and communication parameters. Furthermore, they define the default behaviour of CANopen devices. Despite the strict rules, CANopen device profiles also offer the option of implementing manufacturer-specific device behaviour. Thus system integrators have the option of adding functions on top of the default behaviour.
For most medical devices, the CANopen device profiles define a device finite state automaton (FSA). An FSA specifies device states, corresponding internal and external device behaviour and the events that cause a certain state transition.
So far, a device profile for X-ray collimators (CiA 412-2) has been developed jointly by GE Healthcare, Philips Medical Systems and Siemens Medical Solutions, under the CiA umbrella. The main task of an X-ray collimator is to limit the X-ray beam to a defined format. The device profile supports several functions for focusing the X-ray beam, such as rectangular or circular collimation. Furthermore, the specification makes it possible to support the use of filters for influencing the spectral characteristics of the X-ray beam. The visual simulation of the X-ray beam is functionally incorporated into this device profile too.
For more than 15 years, Philips Medical Systems has used CAN and CANopen as command data communication technology in user interfaces, image detection, X-ray generation, X-ray collimation and the control of the mechanical moving parts of the X-ray system. CANopen is an enabler for the standardisation of component interfaces. Further device profiles for X-ray generators (CiA 412-3), X-ray patient tables (CiA 412-4) and X-ray stands (CiA 412-5) are under development.
Another medical profile, the CANopen device profile for dose measurement systems (CiA 412-6), was developed by CiA members (including Siemens Medical Solutions and PTW). The dose measurement system is used to measure the X-ray dose, the dose area product, the entrance / skin dose and the corresponding dose rates in an X-ray machine. It also measures the irradiation time and factors that influence dose measurement, such as chamber temperature and air pressure. The CANopen device profile defines how the field values (actual measured values) are converted to the desired process values (desired measured value).
Standards for a new class of device profiles for medical diagnostic add-on modules, such as contrast media injectors and Electro-Cardiograms (ECG), are being developed. The application profile for contrast media injectors covers devices used in angiography and those used with CT scanners. The profile also supports multi-piston injectors. The scanner or image-processing device always provides CANopen Manager functionality. The injector device functions as CANopen NMT slave device. To avoid the assignment of node-identifiers, a geographical node-addressing scheme is recommended in the profile.
CANopen networks are now used in many medical and healthcare fields: in laboratory analysis equipment, for example. Medical testing laboratories automate their test procedures to stay competitive and reduce costs, while drug and biotech companies do the same to speed up their development cycles. As a result, automated material handling and storage systems have been introduced.
To standardise a CANopen application profile for laboratory automation, CiA has established a special interest group (SIG). The SIG started with the development of the CiA 434-2 dilutor profile.
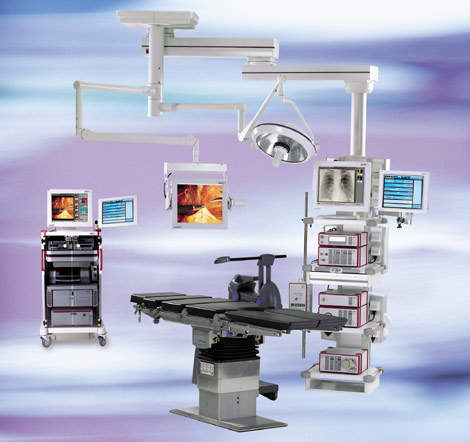
CANopen is also used in several automated Operating Rooms (OR) for endoscope surgery. Besides OR lighting, light projectors, cameras, insufflators (CO2) and HF scalpels have been equipped with CANopen interfaces. Device profiles for these have not yet been standardised, but CiA will do this when the market asks it to.
WORKING TOGETHER
According to Steve Dixon from Hill- Rom, most patients in hospital beds would be amazed if they knew the number of electronic modules required to deliver premium care for patients and improve caregiver effectiveness. Up to 20 independent microprocessors can be communicating together. Just raising a patient’s head can involve the use of motors, several types of sensor and multiple user controls.
Several manufacturers of patient beds for intensive care and OR tables are using CANopen as an embedded network to control various drives and human-machine interfaces. A standardized CANopen interface for beds that will facilitate communication with other intensive care devices as well as OR devices is under discussion.
CiA members are evaluating how existing IEEE 1073 device profiles can be mapped to CANopen. CiA and the HL7 healthcare devices SIG have established a joint task force that will develop a framework for X73-to-CANopen gateways. This task force will also identify applications for combined CANopen / ISO 11073-based networks. In addition, CiA will call for experts to develop dedicated CANopen application profiles for intensive care and automated endoscope surgery systems.
These activities are approved and supervised by CiA’s CANopen medical devices SIG. The development of other application profiles will be carried out as required.





