It is well established that medical devices should be designed to fulfil the requirements of their users.
Research has demonstrated the links between design, usability and human error, and as a result European and US regulators now require manufacturers to demonstrate, through compliance with standards such as ISO 62366, that they have applied user-centred design (UCD), also known as human factors or usability engineering, throughout development.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
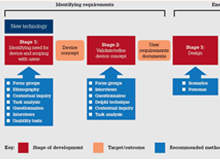
Although designing for patient safety is critically important, satisfying user requirements means considering many other issues including comfort, ease of use, training, storage and labelling. The following user research should be conducted at each stage of development.
Identify a need
Early user research should aim to establish whether there is a clinical need for a new device and identify how that device could make a positive healthcare contribution.
For example, how will the provision of a new device increase patient safety, reduce costs or improve patient outcomes or satisfaction? At this stage we are unlikely to know what all of the relevant issues are so it is important that the scope of the research is as broad as possible.
Users should be encouraged to talk freely about the things that make their life or work difficult, unpleasant or unsafe. The aim should be to fully understand what is it like to treat cancer patients, for example, or to live with Parkinson’s disease.
If some information is already known about this then a more focused study may be appropriate. For example, a surgeon may have identified a problem with a particular procedure and how a specific device could solve it. In this case a detailed study of the procedure would be required using a method such as task analysis, ideally in a number of hospitals in order to gain a complete understanding of how the procedure varies and why.
Validate the device concept
After an initial concept for a device has been identified, a detailed study of the potential users and environments of use should be conducted.
‘Users’ should include not only the target audience who will use the device regularly, but also others who may use it occasionally or help others to use it. For any one device there may be multiple user groups, all exhibiting potential variations in terms of age, educational background, socio-economic group or physical characteristics.
The focus should be on the issues relevant to that specific device. For an assistive device this may be the physical capabilities of patients (and perhaps also their carers), the tasks they perform and the relevant environments.
For an infusion pump, however, the focus would be more on the professional skills and working patterns of clinical users. Understanding user capabilities and characteristics is important for device design, but it will also allow initial and longer-term training needs to be identified. It is important to understand the working patterns of potential users and the clinical pathways and healthcare system of which the device will be a part.
This will allow identification of ‘normal use’, which is necessary to meet medical device regulations.
However, devices will often be used in ways that are not intended by the manufacturer; this may be due to a mistake, but it may also be the result of operational requirements or a clinical emergency. Usability regulations also require that this type of use is investigated and accounted for in design.
The intended outcome of this research is a user requirements document containing the information necessary to produce a prototype design. It can also be used to identify the outcome measures for device evaluation. For example:
- Set-up of the device must take no more than 90 seconds.
- The device should be easy to master by a wide range of users including those without any training.
- The device must be compatible with devices x and y.
- The device must be usable by people with arthritis.
For many devices there will be multiple users with differing specialities, skills and abilities; for example, a device may be used by both professional and lay users. The requirements of these groups must be identified and any conflicts between them resolved. This may mean the requirements of some groups need to be prioritised; for example, untrained users.
Evaluate effectively
A fundamental principle of user-centred design is that it should be iterative and involve a number of cycles of evaluation and re-design. Device evaluation should involve not only safety and usability, but also factors such as functionality, clinical efficacy and cost viability.
It may be tempting to delay user testing until a fully functional prototype is available; however, this can be problematic. If a device has already been CE marked, for example, then even changing the buttons on a user interface will require re-certification, thereby making it too expensive to implement.
User evaluation should be conducted alongside functional evaluations so that feedback can be implemented at the same time. This will mean that early evaluation will normally be performed outside of the clinical environment. The outcome measures of evaluation should be based on the user requirements document.
Example criteria may include:
- improving patient safety by reducing programming errors
- ensuring 90% of users will be able to use the device without training
- reducing the length of a surgical procedure by 15 minutes on average
- enabling patients to remain living independently in their own home.
The aim of early evaluation should be to identify areas for improvement such as:
- The screen must be brighter – it cannot be read in low lighting.
- The device should also measure blood pressure.
- More detailed instructions for the device are required.
- Alarm tone should be changed – it is similar to that of an existing device.
It is important to set the outcome goals at the outset so that the evaluation is objective. As with all stages of development, there is unlikely to be complete agreement between users as to whether all the requirements have been met.
It is important to recognise that what users say they want may not be the same thing as what is safe. It may be necessary, therefore, to sacrifice issues such as speed or ease-of-use for safety.
New usability standards (AAMI/ANSI HE75: ‘Human factors design process for medical devices’) require that the methods and results of user testing are made available to purchasers and users. If testing has been carried out in a simulated clinical environment, for example, then it is important that purchasers are aware of this and the restrictions to the validity of the testing that this may entail. Similarly, if testing has not considered the use of the device by patients then it is important that purchasers are aware of this.
Employ multiple research methods
The choice of methods to be used at each stage of development will depend on a number of issues, including the types of users to be studied and whether the aim is to understand user behaviour or user opinions.
Method choice will also be influenced by whether we are interested in the views of individuals or the views of a group. For example, a focus group discussion can often produce a large number of opinions and experiences. However, it is difficult to effectively manage group discussions, particularly when they contain individuals from different social or professional backgrounds, such as senior surgeons and junior nurses. In addition, individuals may find it difficult to express conflicting opinions or to raise personal or sensitive topics in a group situation.
User research should not rely solely on discursive methods as these only capture information that users are willing to articulate at that particular moment and people often do not behave in the way that they say they do.
Manufacturers should also consider complementing this type of research with observations as this allows user behaviour to be investigated. Contextual inquiry is an effective observational method for identifying dangerous or inefficient behaviours that users are not consciously aware of due to familiarity.
Ideally, more than one method should be applied at each stage to allow different information to be collected. For example, a contextual inquiry may identify information that can then be discussed in depth in an interview or focus group.
Challenge your assumptions
An important purpose of user research is to challenge your assumptions. When closely involved with a project it is common to develop a belief that there is a clear market for a new device and that it will transform patient care. This can lead to an ‘ugly baby syndrome’ where developers do not want to hear negative opinions or feedback.
This is entirely normal; well established cognitive biases mean that we are all predisposed to listen more to information that confirms our beliefs and place less importance upon information that contradicts them. Taking an objective approach to user research is the first step towards ensuring that your new device is entirely safe and fully meets the requirements of your users.
This article is based on ‘Design for patient safety: user testing in the development of medical devices’, published by the UK National Patient Safety Agency and the Multidisciplinary Assessment of Technology Centre for Healthcare.





