The dressings that help the body’s outer armour repair itself are deploying evermore exotic materials to meet the needs of a growing $7.2bn global market.
That growth is being driven by an ageing, less ambulatory population that is more prone to chronic wounds, and the increasing incidence of diabetes with the skin lesions associated with the disease.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Advanced wound care products represent the largest and fastest growing segment of the global market, bringing healthcare providers more options for patient care than ever before. Estimated at $4.1bn in 2006 with a projected annual growth rate of 10%, this category of products includes moist dressings and bandages, antimicrobial dressings incorporating active agents such as silver, and biologically based products incorporating natural growth factors, engineered tissues or skin substitutes.
Collectively, these innovative products account for nearly three fifths of the overall market. Other products and solutions that comprise the broader wound care market include traditional, low-tech bandages or dressings typically made of gauze or non-woven substrates, and non-adherent bandages that are available over the counter.
Devices for wound therapy also continue to make great strides. Non-invasive systems, such as negative pressure systems, hyperbaric oxygen therapies, electrical stimulation, low-level laser therapy and therapeutic ultrasound, have all penetrated the market successfully as viable options. Even exotic therapies such as larvae and maggots or viruses have re-emerged as wound therapies.
Leading the way

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataTechnologically, advanced wound care product development centres
on three factors:
- a moist substrate such as a hydrogel, hydrocolloid, alginate or foam, which provide coverage and protection while remaining ‘breathable’
- the addition of antimicrobials, such as silver, to prevent infection and thus accelerate healing
- the addition of natural epithelial growth factors that trigger tissue layer regrowth.
With the continued emergence of antibiotic resistant bacteria, containing the wound is critical to preventing infection. Reduction in wound healing time significantly decreases the ongoing costs for wound care, and this will be seen as the pre-eminent driver for advanced wound care technologies. From a broader healthcare perspective, insurance reimbursement decisions are often based primarily on product cost and not necessarily on improvements in wound healing time and subsequent quality of life.
Home-based products and solutions can address cost issues by reducing the number of doctor’s visits, but with any home-based therapies, proper use and patient compliance are critical in determining outcomes. In the regulatory arena, many of the advanced wound care products are designated as combination products; they incorporate a device with a therapeutic agent (pharmaceutical or biological) to deliver a single therapeutic action. Anti-microbial silver dressings are an example of this technology.
The US FDA has led the way in establishing guidelines for reviewing combination products in terms of their definition, designation of primary mode of action, designation of jurisdiction for review and approval of submissions, and post-market surveillance. The FDA established its Office of Combination Products in 2002 to handle the increasingly complex products submitted for market approval.
The office determines the primary mode of action, or which component is most responsible for delivering the therapeutic agent or application. Consequently, the jurisdiction for primary review and approval will be designated either to the Center for Devices and Radiological Health (CDRH), the Center for Drug Evaluation and Research (CDER), or the Center for Biologics Evaluation and Research (CBER). This ensures that effective reviews can be conducted through the appropriate agency, while other offices can be integrated for review as necessary.
While the US has led the way in terms of regulatory guidelines for combination products such as advanced wound care, the UK and Europe have lagged behind in instituting more definitive pathways for approvals.
Regulatory agencies in the UK and Europe must identify and implement a reasonable and effective approach for reviewing products based upon separate technologies. The UK Healthcare Industry Task Force Review (HITF) has already recognised this challenge. In the EU the regulatory path for reviewing market submissions for novel combination products is unclear because the European Medicines Agency (EMEA) is not responsible for regulatory oversight of medical devices. As more advanced wound care products are approved for market either in the US or Europe, regulatory agencies will gain more knowledge and expertise in handling these submissions.
This will enhance the willingness of companies to develop advanced wound care products.
Moist wound dressings
Moist wound dressings use materials such as hydrogels, alginates, or hydrocolloids to accelerate healing. The moist environment created by such dressings reduces tissue adhesion to the dressing and lessens trauma upon removal. While these products dominate the advanced wound care market, their share appears to be levelling off due to the maturity of the market. Patent applications and granted patents have declined in the past few years.
Active wound products
Active products incorporate engineered tissue such as collagen and epidermal growth factors. Ethicon’s Promogran dressing, for example, is constructed of collagen and regenerated cellulose (ORC). It inhibits matrix metalloproteases (MMPs), enzymes that may have a negative effect on wound healing. Products such as these open opportunities for pharmaceutical and biotechnology companies to collaborate with the medical device industry, which might lead to even more treatment options. For example, LifeCell Corporation is developing active wound care technologies in conjunction with Boston Scientific, Wright Medical, Stryker Corporation, and BioHorizons.
Other technologies
Other non-tissue-assisted closure systems represent a non-invasive approach to wound healing. According to a recent report by Research and Markets, KCI is now the market leader in advanced wound care solutions with the success of this type of system. Devices for wound therapy include hyperbaric technologies such as Kinetic Concepts’ negative pressure, vacuum- oxygen, electrical stimulation, low-level laser therapy, and therapeutic ultrasound has also successfully penetrated the wound care market. These systems are non-invasive, rapid and effective, and can be employed on an out-patient basis, making them preferable to the provider, insurer and patient.
Moist wound care: the benefits
As newer technologies are developed, moist wound care products are lessening in popularity. However, they still have positive treatment benefits for many.
Hydrogels. Hydrogels are natural or synthetic cross-linked polymers used in a variety of medical and biomedical applications. They are used in the construction of contact lenses, drug-delivery vehicles, and as physiological electrodes or sensors. Hydrogels also exhibit excellent skin adhesion and high moisture vapour transmission properties. A hydrogel is a three-dimensional polymer network that contains water, giving it a high degree of hydrophilicity while remaining insoluble. The main component is based on water-soluble monomers. The extent of hydrophilicity can be modified by copolymerisation with monomers of varying degrees of hydrophobicity and hydrophilicity. In addition to the water and polymer content, they may contain a plasticiser that facilitates the matrix formulation for good adhesion properties.
Hydrocolloids. Hydrocolloid dressings contain gel-forming agents such as sodium carboxymethylcellulose (NaCMC) and gelatin. They are often combined with elastomeric materials into a polyurethane foam or film matrix. Like all moist dressings they can be left in place for two to five days, maintaining a moist environment while minimising tissue adhesion to the dressing. They also exhibit good absorbency of wound exudates.
Alginates. Alginates are formulated from natural chitosan, a proteinaceous component of seaweed. Their excellent absorption properties are achieved via strong hydrophilic gel formations that limit wound exudates and minimise bacterial contamination. Alginate fibres are biodegradable and elicit little or no allergic reaction as compared to synthetic dressings.
Antimicrobials. Silver has long demonstrated effective antimicrobial properties and has been widely used in dental and other antibiotic applications. Silver appears to promote skin regrowth, and it may even possess anti-inflammatory capacities. It also has demonstrated effectiveness against antibiotic-resistant strains such as methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant Enterococcus (VRE).
This is a critical property given the rise in antibiotic-resistant bacterial infections, particularly MRSA. According to estimates from the US Centers for Disease Control and Prevention, more than 94,500 cases of invasive MRSA were reported in 2005. Approximately 85% of these are hospital- or healthcare-associated infections, with a majority of these occurring in elderly populations.
Market leaders
Four companies dominate the overall wound care market. Smith & Nephew still leads in moist dressings, while Johnson & Johnson, 3M, and Beiersdorf AG have large shares of the advanced wound care market. Numerous smaller innovative companies, such as Hollister, Carrington Labs, O’Classen Pharmaceuticals, Molnlycke, Kendall and C R Bard, have either successfully penetrated this market with their own product lines or are licensing or partnering with the larger players.
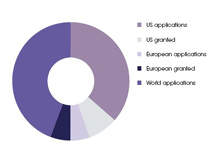
Figure 1 shows the top companies based on worldwide 2007 patent activity, either granted patents or applications for advanced wound care products. Approximately 350 patents or applications were published in 2007 for these technologies.
Wound care technology adoption
The acceptance of advanced wound care solutions is reported to have been slower in the UK and Europe due to differences in cost-benefit analyses. In the US, health insurers may consider not only the cost of the device but the also the longer-term cost savings to the patient and to the healthcare system. In the UK and Europe, cost considerations for reimbursement might focus only on the cost of the device itself.
Another factor influencing the acceptance of advanced wound care products in the UK and Europe is that wound management is often facilitated by nurses, as opposed to more specialised physicians in the US. As many of the advanced wound care products include biological tissues or growth factors, it is imperative to have trained personnel with skill in handling these products.
The overall wound care market in the US is estimated at $20bn annually, with more than $4bn spent on products like dressings. Aside from the various product designs, the wound management market is driven and characterised by various medical conditions and indications. Skin or pressure ulcers are prevalent in elderly populations and are also a common sequela of diabetes. The incidences of diabetes are forecast to increase 165%, affecting up to 29 million people in the US alone by 2050. Also, the percentage of Americans over 65 is expected to grow faster than the total population in the US up to 2030. Advanced wound care products described in this article are the preferred treatments for these indications.
The average hospital stay has decreased from 6.4 days in 1990 to 4.6 days in 2003, and the number of surgeries performed in ambulatory surgical centres increased 191% between 1990 and 2000, indicating a need for products that accelerate wound healing. These trends indicate a growing need for not only advanced wound care products but also home-based care solutions.
Home-based or outpatient care is driven by the ageing population and concurrent skin ulcers associated with diabetes and/or immobility. Pressure ulcers, skin ulcers and diabetic foot ulcers are the most prevalent conditions within this population. Moist dressings appear to be the most valuable treatment option based on their effective wound healing and infection prevention.
These dressings are also covered and reimbursed by the major healthcare providers and government associations such as Medicare/Medicaid. Ostomy site care is also a significant condition in the ageing population, requiring home-based care to prevent infections.
Nursing practices are impacted by the shift towards home care, not just in terms of on-site care and support, but also by the use of web-based technologies for remote monitoring and evaluation. A continued emphasis on developing and implementing best practices for home-based care of wounds is a great challenge for nurses.





