
For healthcare systems working to improve clinical outcomes for the millions of people who suffer from strokes every year, the key issue is time. Every minute that can be saved during a cerebral vascular accident can have a significant impact on the chance and severity of permanent brain damage and disability. From onset to treatment, the US National Institute of Neurological Disorders and Stroke recommends a ‘door-to-needle’ time of one hour or less, a timescale that many hospitals and medical centres struggle to meet.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
In the case of ischaemic stroke, which is by far the most common form, treatment takes the form of thrombolysis or mechanical thrombectomy. These work to either break down or remove clots blocking the flow of blood to the brain. However, rapidly interpreting complex computed tomography (CT) scan data to identify the right treatment for a given patient is a tough task, especially for clinicians who aren’t specialist neuroradiologists.
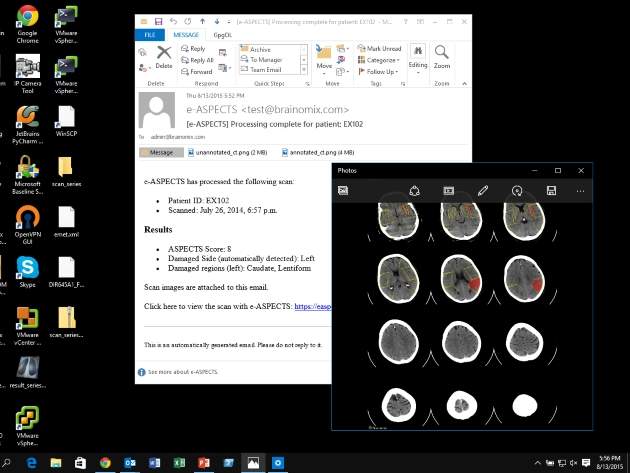
Oxford University spin-out Brainomix is making a new contribution to the time-to-treatment dilemma inherent to stroke cases. Its e-ASPECTS software, which has the European CE Mark as a Class 2a medical device and is in use in Brazil and a number of countries across Europe, is an artificial intelligence-driven system that interprets CT scans to generate an automated ASPECTS score. The ASPECTS score was invented by Oxford University professor of stroke medicine and Brainomix co-founder Alastair Buchan. It provides a quantitative CT scan assessment method that uses observations of ten key regions of the brain to arrive at a score of ‘0’ (most severe) to ’10’ (least severe).
The ASPECTS score has gained traction in the medical field, proving especially useful for clinical researchers, who have used it to select stroke patients who are likely to benefit from new treatments. However, it can be too difficult to use under pressure in a public health setting. e-ASPECTS is designed to quickly generate an automated score that can support a clinician’s decision-making during this all-important ‘door-to-needle’ moment. US Food and Drug Administration (FDA) approval is pending for use in the US, with a North American launch of the software tentatively expected next year.
Brainomix chief technology officer Eric Greveson gives more detail about e-ASPECTS and the Angels Initiative that aims to accelerate stroke diagnosis across Europe.
Chris Lo: Where is e-ASPECTS currently being used?
EG: We got the CE Mark as a Class 2a medical device a couple of years ago, so I guess that was in the beginning of 2015. Following that, we had some initial installations in the UK and Germany, and also in Finland, where we got some initial feedback on how the software works and made some minor developments. But since then we’ve been rolling it out across Europe. So now we’ve got installations in many European countries.
That’s been accelerating recently because we’ve partnered with the Angels Initiative. In addition to the territories we’ve already been involved in – so the Nordics, Germany, the UK; we’ve now got a lot of countries and central and southern Europe that are installing e-ASPECTS in their hospitals.
CL: What role does artificial intelligence play in the software’s ability to reliably automate the ASPECTS scoring system for CT scans?
EG: It does not have access to the clinical information, so it’s just trying to work out what’s going on from the image itself. With lots of image analysis software, and e-ASPECTS is the same, we use advanced machine learning classifiers to look at image features and detect things that are abnormal, based on a training data set, essentially. So what we do is train the software to look for signs of ischaemia and stroke damage, and then if we’ve got a large enough data set, the machine can learn how to do that, and then expend it on new data sets that it hasn’t seen before. That’s what we mean by artificial intelligence.
CL: How important has it been to have Professor Alastair Buchan, the inventor of the ASPECTS score, as a co-founder and senior medical science advisor to help guide the development of your software?
EG: It’s been really useful. Having somebody who knows the whole history of ASPECTS and why it was invented was very useful, especially at the start when we started developing the software. In addition to his clinical contacts, he can provide feedback and advice on what the radiologists and neurologists are looking for in the software.
Another important point is that Michalis Papadakis, who is the CEO of the company, was actually working for Alastair before we started the company. Michalis was scientific director of the preclinical stroke lab in Oxford, and it was he and Alastair who came up with the idea of automating ASPECTS.
CL: A study was recently published suggesting that e-ASPECTS can act as a predictor for the outcomes of mechanical thrombectomy in acute ischemic stroke patients – are you looking into other stroke imaging procedures that the software could support?
EG: One thing that we’re working on at the moment is other modalities, so not just plain CT. We’re working on a couple of projects that haven’t been announced yet, but no doubt we’ll be announcing something soon, in the next few months, where we’re looking at expanding it beyond just plain CT and starting to work out the other modalities that are also commonly used in stroke imaging. Our overall aim is to provide a suite of software that helps with all of the different imaging that would be related to a patient’s case.
CL: What was behind the decision to partner with Boehringer Ingelheim to offer six months of free usage of the software to hospitals participating in the Angels Initiative?
EG: The first thing to mention is that the Angels Initiative was a huge opportunity for us. We’re still a relatively small company, and we can’t easily employ staff in many different countries. But what the Angels Initiative gave us is a way to start rolling out e-ASPECTS in all these European countries, with the introductions to hospitals being provided through the initiative. So it’s really accelerated our rollout in all of these countries.
As far as the benefits to physicians and patients go, it actually varies from country to country, because some countries have more advanced and integrated stroke systems than others. The main benefits are saving time for the physicians, but also having more understanding of the quantitative assessment of CT scans and getting more of the right patients selected for the right treatment, even after hours or when there aren’t expert neuroradiologists available.
CL: As the Angels Initiative moves forward, what is the future scenario you are hoping for, in terms of improving stroke diagnosis and treatment across Europe?
EG: The overall objective of the Angels Initiative, even for those hospitals that don’t have e-ASPECTS, is to speed up the stroke treatment pathway, to reduce the time from onset to treatment and make sure that the right patients are being selected for those treatments.
CL: e-ASPECTS has launched in Brazil – why was Brazil the first country outside of Europe to be introduced to the software?
EG: We’re actually part of the RESILIENT trial in Brazil. The RESILIENT trial is probably going to be the last mechanical thrombectomy trial. The reason they’re doing another trial even though the existing thrombectomy trials have shown it to be a beneficial treatment is because all those trials were in developed, western-style healthcare systems.
The RESILIENT trial is to show that mechanical thrombectomy can work even in a developing country like Brazil, which might not have the same resources and is dealing with difficult geography certainly, and they want to show that you can still have a benefit to patients. So all of South America is looking to this trial to see if it’s going to work before they decide to implement mechanical thrombectomy.
CL: What are the economics of e-ASPECTS for health systems with tight budgets?
EG: When we have to make a health economic case to a hospital, we have to demonstrate that e-ASPECTS will be good value for money for them. That often means making savings through faster treatment, through slightly improved patient outcomes, which can lead to reduced bed days, or through selecting more of the right patients for the right treatment.
With modern hospital networks and so on, you can even have one installation that can work for several hospitals if you’ve got rapid network connections between them. So we don’t have to install any physical hardware. In some places we still do, especially if the hospital doesn’t have a very modern IT infrastructure, but in most hospitals they can support virtualised machines, and we can then install e-ASPECTS, even remotely, and save a lot of time and cost.



